[English] 日本語
 Yorodumi
Yorodumi- EMDB-41577: mGluR3 in the presence of the antagonist LY 341495 and positive a... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | mGluR3 in the presence of the antagonist LY 341495 and positive allosteric modulator VU6023326 | ||||||||||||
 Map data Map data | Full length map | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | GPCR / synaptic protein / MEMBRANE PROTEIN | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationClass C/3 (Metabotropic glutamate/pheromone receptors) / group II metabotropic glutamate receptor activity / G protein-coupled glutamate receptor signaling pathway / G alpha (i) signalling events / astrocyte projection / cellular response to stress / postsynaptic modulation of chemical synaptic transmission / regulation of synaptic transmission, glutamatergic / sensory perception of pain / calcium channel regulator activity ...Class C/3 (Metabotropic glutamate/pheromone receptors) / group II metabotropic glutamate receptor activity / G protein-coupled glutamate receptor signaling pathway / G alpha (i) signalling events / astrocyte projection / cellular response to stress / postsynaptic modulation of chemical synaptic transmission / regulation of synaptic transmission, glutamatergic / sensory perception of pain / calcium channel regulator activity / modulation of chemical synaptic transmission / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / presynaptic membrane / scaffold protein binding / gene expression / dendritic spine / postsynaptic membrane / neuron projection / postsynaptic density / axon / glutamatergic synapse / plasma membrane Similarity search - Function | ||||||||||||
| Biological species |  | ||||||||||||
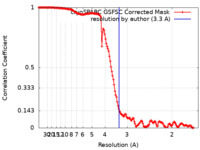
| Method | single particle reconstruction / cryo EM / Resolution: 3.3 Å | ||||||||||||
 Authors Authors | Strauss A / Levitz J | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: bioRxiv / Year: 2023 Journal: bioRxiv / Year: 2023Title: Structural basis of allosteric modulation of metabotropic glutamate receptor activation and desensitization. Abstract: The metabotropic glutamate receptors (mGluRs) are neuromodulatory family C G protein coupled receptors which assemble as dimers and allosterically couple extracellular ligand binding domains (LBDs) ...The metabotropic glutamate receptors (mGluRs) are neuromodulatory family C G protein coupled receptors which assemble as dimers and allosterically couple extracellular ligand binding domains (LBDs) to transmembrane domains (TMDs) to drive intracellular signaling. Pharmacologically, mGluRs can be targeted either at the LBDs by glutamate and synthetic orthosteric compounds or at the TMDs by allosteric modulators. Despite the potential of allosteric TMD-targeting compounds as therapeutics, an understanding of the functional and structural basis of their effects on mGluRs is limited. Here we use a battery of approaches to dissect the distinct functional and structural effects of orthosteric versus allosteric ligands. We find using electrophysiological and live cell imaging assays that both agonists and positive allosteric modulators (PAMs) can drive activation and desensitization of mGluRs. The effects of PAMs are pleiotropic, including both the ability to boost the maximal response to orthosteric agonists and to serve independently as desensitization-biased agonists across mGluR subtypes. Conformational sensors reveal PAM-driven inter-subunit re-arrangements at both the LBD and TMD. Motivated by this, we determine cryo-electron microscopy structures of mGluR3 in the presence of either an agonist or antagonist alone or in combination with a PAM. These structures reveal PAM-driven re-shaping of intra- and inter-subunit conformations and provide evidence for a rolling TMD dimer interface activation pathway that controls G protein and beta-arrestin coupling. HIGHLIGHTS: -Agonists and PAMs drive mGluR activation, desensitization, and endocytosis-PAMs are desensitization-biased and synergistic with agonists-Four combinatorial ligand conditions reveal an ...HIGHLIGHTS: -Agonists and PAMs drive mGluR activation, desensitization, and endocytosis-PAMs are desensitization-biased and synergistic with agonists-Four combinatorial ligand conditions reveal an ensemble of full-length mGluR structures with novel interfaces-Activation and desensitization involve rolling TMD interfaces which are re-shaped by PAM. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41577.map.gz emd_41577.map.gz | 136.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41577-v30.xml emd-41577-v30.xml emd-41577.xml emd-41577.xml | 30.3 KB 30.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_41577_fsc.xml emd_41577_fsc.xml | 13.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_41577.png emd_41577.png | 44.5 KB | ||
| Filedesc metadata |  emd-41577.cif.gz emd-41577.cif.gz | 6.8 KB | ||
| Others |  emd_41577_additional_1.map.gz emd_41577_additional_1.map.gz emd_41577_additional_2.map.gz emd_41577_additional_2.map.gz emd_41577_additional_3.map.gz emd_41577_additional_3.map.gz emd_41577_additional_4.map.gz emd_41577_additional_4.map.gz emd_41577_additional_5.map.gz emd_41577_additional_5.map.gz emd_41577_additional_6.map.gz emd_41577_additional_6.map.gz emd_41577_half_map_1.map.gz emd_41577_half_map_1.map.gz emd_41577_half_map_2.map.gz emd_41577_half_map_2.map.gz | 196.9 MB 196.9 MB 137.1 MB 128.8 MB 254.9 MB 254.9 MB 254.4 MB 254.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41577 http://ftp.pdbj.org/pub/emdb/structures/EMD-41577 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41577 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41577 | HTTPS FTP |
-Related structure data
| Related structure data |  8trcMC  8tqbC  8tr0C  8tr2C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_41577.map.gz / Format: CCP4 / Size: 274.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41577.map.gz / Format: CCP4 / Size: 274.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Full length map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.825 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Half map A of the transmembrane domain following particle subtraction
| File | emd_41577_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map A of the transmembrane domain following particle subtraction | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Half map B of the transmembrane domain following particle subtraction
| File | emd_41577_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map B of the transmembrane domain following particle subtraction | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Locally refined map of the extracellular domain
| File | emd_41577_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Locally refined map of the extracellular domain | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Map of the transmembrane domain following particle subtraction
| File | emd_41577_additional_4.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Map of the transmembrane domain following particle subtraction | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Half map A for the locally refined extracellular domain
| File | emd_41577_additional_5.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map A for the locally refined extracellular domain | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Half map B for the locally refined extracellular domain
| File | emd_41577_additional_6.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map B for the locally refined extracellular domain | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map A for full length map
| File | emd_41577_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map A for full length map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map B for full length map
| File | emd_41577_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map B for full length map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Metabotropic Glutamate Receptor 3 dimer
| Entire | Name: Metabotropic Glutamate Receptor 3 dimer |
|---|---|
| Components |
|
-Supramolecule #1: Metabotropic Glutamate Receptor 3 dimer
| Supramolecule | Name: Metabotropic Glutamate Receptor 3 dimer / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 260 KDa |
-Macromolecule #1: Metabotropic glutamate receptor 3
| Macromolecule | Name: Metabotropic glutamate receptor 3 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 103.161555 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MKMLTRLQIL MLALFSKGFL LSLGDHNFMR REIKIEGDLV LGGLFPINEK GTGTEECGRI NEDRGIQRLE AMLFAIDEIN KDNYLLPGV KLGVHILDTC SRDTYALEQS LEFVRASLTK VDEAEYMCPD GSYAIQENIP LLIAGVIGGS YSSVSIQVAN L LRLFQIPQ ...String: MKMLTRLQIL MLALFSKGFL LSLGDHNFMR REIKIEGDLV LGGLFPINEK GTGTEECGRI NEDRGIQRLE AMLFAIDEIN KDNYLLPGV KLGVHILDTC SRDTYALEQS LEFVRASLTK VDEAEYMCPD GSYAIQENIP LLIAGVIGGS YSSVSIQVAN L LRLFQIPQ ISYASTSAKL SDKSRYDYFA RTVPPDFYQA KAMAEILRFF NWTYVSTVAS EGDYGETGIE AFEQEARLRN IC IATAEKV GRSNIRKSYD SVIRELLQKP NARVVVLFMR SDDSRELIAA ANRVNASFTW VASDGWGAQE SIVKGSEHVA YGA ITLELA SHPVRQFDRY FQSLNPYNNH RNPWFRDFWE QKFQCSLQNK RNHRQVCDKH LAIDSSNYEQ ESKIMFVVNA VYAM AHALH KMQRTLCPNT TKLCDAMKIL DGKKLYKEYL LKINFTAPFN PNKGADSIVK FDTFGDGMGR YNVFNLQQTG GKYSY LKVG HWAETLSLDV DSIHWSRNSV PTSQCSDPCA PNEMKNMQPG DVCCWICIPC EPYEYLVDEF TCMDCGPGQW PTADLS GCY NLPEDYIKWE DAWAIGPVTI ACLGFLCTCI VITVFIKHNN TPLVKASGRE LCYILLFGVS LSYCMTFFFI AKPSPVI CA LRRLGLGTSF AICYSALLTK TNCIARIFDG VKNGAQRPKF ISPSSQVFIC LGLILVQIVM VSVWLILETP GTRRYTLP E KRETVILKCN VKDSSMLISL TYDVVLVILC TVYAFKTRKC PENFNEAKFI GFTMYTTCII WLAFLPIFYV TSSDYRVQT TTMCISVSLS GFVVLGCLFA PKVHIVLFQP QKNVVTHRLH LNRFSVSGTA TTYSQSSAST YVPTVCNGRE VLDSTTSSLA GLVPRGSAA AKSAWSHPQF EKGGGSGGGS GGGSWSHPQF EK UniProtKB: Metabotropic glutamate receptor 3 |
-Macromolecule #2: 2-[(1S,2S)-2-carboxycyclopropyl]-3-(9H-xanthen-9-yl)-D-alanine
| Macromolecule | Name: 2-[(1S,2S)-2-carboxycyclopropyl]-3-(9H-xanthen-9-yl)-D-alanine type: ligand / ID: 2 / Number of copies: 2 / Formula: Z99 |
|---|---|
| Molecular weight | Theoretical: 353.369 Da |
| Chemical component information | 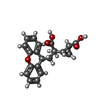 ChemComp-Z99: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation #1
Sample preparation #1
| Preparation ID | 1 |
|---|---|
| Concentration | 4.5 mg/mL |
| Buffer | pH: 7.5 |
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Film type ID: 1 / Support film - Material: GOLD / Support film - topology: HOLEY / Pretreatment - Type: PLASMA CLEANING |
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
- Sample preparation #2
Sample preparation #2
| Preparation ID | 2 |
|---|---|
| Buffer | pH: 7.5 |
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Film type ID: 1 / Support film - Material: GOLD / Support film - topology: HOLEY / Pretreatment - Type: PLASMA CLEANING |
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Detector mode: COUNTING / Average electron dose: 58.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.7000000000000001 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-8trc: |
 Movie
Movie Controller
Controller












 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)