[English] 日本語
 Yorodumi
Yorodumi- EMDB-3842: Structure of a native assembly intermediate of the human mitochon... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-3842 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of a native assembly intermediate of the human mitochondrial ribosome with unfolded interfacial rRNA | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of mitochondrial translation / mitochondrial large ribosomal subunit assembly / Complex I biogenesis / protein lipoylation / negative regulation of ribosome biogenesis / Mitochondrial Fatty Acid Beta-Oxidation / Protein lipoylation / rRNA import into mitochondrion / mitochondrial [2Fe-2S] assembly complex / Respiratory electron transport ...negative regulation of mitochondrial translation / mitochondrial large ribosomal subunit assembly / Complex I biogenesis / protein lipoylation / negative regulation of ribosome biogenesis / Mitochondrial Fatty Acid Beta-Oxidation / Protein lipoylation / rRNA import into mitochondrion / mitochondrial [2Fe-2S] assembly complex / Respiratory electron transport / mitochondrial translational termination / mitochondrial translational elongation / translation release factor activity, codon nonspecific / positive regulation of mitochondrial translation / microprocessor complex / Mitochondrial translation elongation / Mitochondrial translation termination / Mitochondrial translation initiation / iron-sulfur cluster assembly complex / mitochondrial large ribosomal subunit binding / mitochondrial fission / mitochondrial large ribosomal subunit / Hydrolases; Acting on ester bonds; Endoribonucleases producing 5'-phosphomonoesters / peptidyl-tRNA hydrolase / mitochondrial small ribosomal subunit / [2Fe-2S] cluster assembly / aminoacyl-tRNA hydrolase activity / respiratory chain complex I / mitochondrial ribosome / iron-sulfur cluster assembly / mitochondrial translation / ribosomal large subunit binding / proton motive force-driven mitochondrial ATP synthesis / : / mitochondrial electron transport, NADH to ubiquinone / mitochondrial respiratory chain complex I assembly / acyl binding / anatomical structure morphogenesis / acyl carrier activity / RNA processing / Mitochondrial protein degradation / aerobic respiration / rescue of stalled ribosome / ribosomal large subunit biogenesis / cellular response to leukemia inhibitory factor / fatty acid binding / mitochondrial membrane / fibrillar center / fatty acid biosynthetic process / double-stranded RNA binding / small ribosomal subunit rRNA binding / cell junction / large ribosomal subunit rRNA binding / 5S rRNA binding / endonuclease activity / mitochondrial inner membrane / negative regulation of translation / nuclear body / rRNA binding / ribosome / mitochondrial matrix / structural constituent of ribosome / ribonucleoprotein complex / translation / protein domain specific binding / mRNA binding / nucleotide binding / calcium ion binding / synapse / nucleolus / apoptotic process / mitochondrion / RNA binding / extracellular space / nucleoplasm / nucleus / plasma membrane / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  Human (human) Human (human) | |||||||||
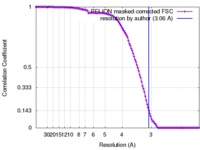
| Method | single particle reconstruction / cryo EM / Resolution: 3.06 Å | |||||||||
 Authors Authors | Brown A / Rathore S / Kimanius D / Aibara S / Bai XC / Rorbach J / Amunts A / Ramakrishnan V | |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2017 Journal: Nat Struct Mol Biol / Year: 2017Title: Structures of the human mitochondrial ribosome in native states of assembly. Authors: Alan Brown / Sorbhi Rathore / Dari Kimanius / Shintaro Aibara / Xiao-Chen Bai / Joanna Rorbach / Alexey Amunts / V Ramakrishnan /   Abstract: Mammalian mitochondrial ribosomes (mitoribosomes) have less rRNA content and 36 additional proteins compared with the evolutionarily related bacterial ribosome. These differences make the assembly of ...Mammalian mitochondrial ribosomes (mitoribosomes) have less rRNA content and 36 additional proteins compared with the evolutionarily related bacterial ribosome. These differences make the assembly of mitoribosomes more complex than the assembly of bacterial ribosomes, but the molecular details of mitoribosomal biogenesis remain elusive. Here, we report the structures of two late-stage assembly intermediates of the human mitoribosomal large subunit (mt-LSU) isolated from a native pool within a human cell line and solved by cryo-EM to ∼3-Å resolution. Comparison of the structures reveals insights into the timing of rRNA folding and protein incorporation during the final steps of ribosomal maturation and the evolutionary adaptations that are required to preserve biogenesis after the structural diversification of mitoribosomes. Furthermore, the structures redefine the ribosome silencing factor (RsfS) family as multifunctional biogenesis factors and identify two new assembly factors (L0R8F8 and mt-ACP) not previously implicated in mitoribosomal biogenesis. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_3842.map.gz emd_3842.map.gz | 15.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-3842-v30.xml emd-3842-v30.xml emd-3842.xml emd-3842.xml | 72.4 KB 72.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_3842_fsc.xml emd_3842_fsc.xml | 12.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_3842.png emd_3842.png | 271.3 KB | ||
| Masks |  emd_3842_msk_1.map emd_3842_msk_1.map | 166.4 MB |  Mask map Mask map | |
| Others |  emd_3842_half_map_1.map.gz emd_3842_half_map_1.map.gz emd_3842_half_map_2.map.gz emd_3842_half_map_2.map.gz | 130.9 MB 131 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-3842 http://ftp.pdbj.org/pub/emdb/structures/EMD-3842 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3842 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3842 | HTTPS FTP |
-Validation report
| Summary document |  emd_3842_validation.pdf.gz emd_3842_validation.pdf.gz | 646.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_3842_full_validation.pdf.gz emd_3842_full_validation.pdf.gz | 645.8 KB | Display | |
| Data in XML |  emd_3842_validation.xml.gz emd_3842_validation.xml.gz | 19.9 KB | Display | |
| Data in CIF |  emd_3842_validation.cif.gz emd_3842_validation.cif.gz | 26.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3842 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3842 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3842 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3842 | HTTPS FTP |
-Related structure data
| Related structure data |  5oolMC  3843C  5oomC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_3842.map.gz / Format: CCP4 / Size: 166.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_3842.map.gz / Format: CCP4 / Size: 166.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.06 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_3842_msk_1.map emd_3842_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_3842_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_3842_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Mitochondrial ribosome large subunit with assembly factor
+Supramolecule #1: Mitochondrial ribosome large subunit with assembly factor
+Macromolecule #1: 16S ribosomal RNA
+Macromolecule #2: mitochondrial tRNAVal
+Macromolecule #3: 39S ribosomal protein L2, mitochondrial
+Macromolecule #4: 39S ribosomal protein L3, mitochondrial
+Macromolecule #5: 39S ribosomal protein L4, mitochondrial
+Macromolecule #6: 39S ribosomal protein L9, mitochondrial
+Macromolecule #7: 39S ribosomal protein L10, mitochondrial
+Macromolecule #8: 39S ribosomal protein L11, mitochondrial
+Macromolecule #9: 39S ribosomal protein L13, mitochondrial
+Macromolecule #10: 39S ribosomal protein L14, mitochondrial
+Macromolecule #11: 39S ribosomal protein L15, mitochondrial
+Macromolecule #12: 39S ribosomal protein L16, mitochondrial
+Macromolecule #13: 39S ribosomal protein L17, mitochondrial
+Macromolecule #14: 39S ribosomal protein L18, mitochondrial
+Macromolecule #15: 39S ribosomal protein L19, mitochondrial
+Macromolecule #16: 39S ribosomal protein L20, mitochondrial
+Macromolecule #17: 39S ribosomal protein L21, mitochondrial
+Macromolecule #18: 39S ribosomal protein L22, mitochondrial
+Macromolecule #19: 39S ribosomal protein L23, mitochondrial
+Macromolecule #20: 39S ribosomal protein L24, mitochondrial
+Macromolecule #21: 39S ribosomal protein L27, mitochondrial
+Macromolecule #22: 39S ribosomal protein L28, mitochondrial
+Macromolecule #23: 39S ribosomal protein L47, mitochondrial
+Macromolecule #24: 39S ribosomal protein L30, mitochondrial
+Macromolecule #25: 39S ribosomal protein L32, mitochondrial
+Macromolecule #26: 39S ribosomal protein L33, mitochondrial
+Macromolecule #27: 39S ribosomal protein L34, mitochondrial
+Macromolecule #28: 39S ribosomal protein L35, mitochondrial
+Macromolecule #29: 39S ribosomal protein L36, mitochondrial
+Macromolecule #30: 39S ribosomal protein L37, mitochondrial
+Macromolecule #31: 39S ribosomal protein L38, mitochondrial
+Macromolecule #32: 39S ribosomal protein L39, mitochondrial
+Macromolecule #33: 39S ribosomal protein L40, mitochondrial
+Macromolecule #34: 39S ribosomal protein L41, mitochondrial
+Macromolecule #35: 39S ribosomal protein L42, mitochondrial
+Macromolecule #36: 39S ribosomal protein L43, mitochondrial
+Macromolecule #37: 39S ribosomal protein L44, mitochondrial
+Macromolecule #38: 39S ribosomal protein L45, mitochondrial
+Macromolecule #39: 39S ribosomal protein L46, mitochondrial
+Macromolecule #40: 39S ribosomal protein L48, mitochondrial
+Macromolecule #41: 39S ribosomal protein L49, mitochondrial
+Macromolecule #42: 39S ribosomal protein L50, mitochondrial
+Macromolecule #43: 39S ribosomal protein L51, mitochondrial
+Macromolecule #44: 39S ribosomal protein L52, mitochondrial
+Macromolecule #45: 39S ribosomal protein L53, mitochondrial
+Macromolecule #46: 39S ribosomal protein L54, mitochondrial
+Macromolecule #47: 39S ribosomal protein L55, mitochondrial
+Macromolecule #48: Ribosomal protein 63, mitochondrial
+Macromolecule #49: Peptidyl-tRNA hydrolase ICT1, mitochondrial
+Macromolecule #50: Growth arrest and DNA damage-inducible proteins-interacting protein 1
+Macromolecule #51: 39S ribosomal protein S18a, mitochondrial
+Macromolecule #52: 39S ribosomal protein S30, mitochondrial
+Macromolecule #53: Unknown protein or protein extension
+Macromolecule #54: Mitochondrial assembly of ribosomal large subunit protein 1
+Macromolecule #55: MIEF1 upstream open reading frame protein
+Macromolecule #56: Acyl carrier protein, mitochondrial
+Macromolecule #57: MAGNESIUM ION
+Macromolecule #58: ZINC ION
+Macromolecule #59: 4'-PHOSPHOPANTETHEINE
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.45 Component:
Details: 2 mM Synercid (Santa Cruz Biotechnology, Inc) was added to reduce preferential orientation. | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 3.0 nm / Pretreatment - Type: GLOW DISCHARGE | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV / Details: Sample incubated for 30 s prior to freezing.. | |||||||||||||||
| Details | Sample concentration = 100 nM |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) / Detector mode: INTEGRATING / Digitization - Frames/image: 1-25 / Average exposure time: 1.5 sec. / Average electron dose: 1.56 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 130841 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller






























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)