+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Structure of mammalian spectrin-actin junctional complex of membrane skeleton, State II, Global map | |||||||||
 マップデータ マップデータ | ||||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Macrocomplex / membrane skeleton / spectrin-actin junction / MEMBRANE PROTEIN | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報regulation of cellular component size / pointed-end actin filament capping / endoplasmic reticulum tubular network organization / negative regulation of protein targeting to membrane / regulation of multicellular organismal development / positive regulation of developmental process / spectrin / lens fiber cell development / Regulation of actin dynamics for phagocytic cup formation / EPHB-mediated forward signaling ...regulation of cellular component size / pointed-end actin filament capping / endoplasmic reticulum tubular network organization / negative regulation of protein targeting to membrane / regulation of multicellular organismal development / positive regulation of developmental process / spectrin / lens fiber cell development / Regulation of actin dynamics for phagocytic cup formation / EPHB-mediated forward signaling / Adherens junctions interactions / VEGFA-VEGFR2 Pathway / Cell-extracellular matrix interactions / RHO GTPases Activate WASPs and WAVEs / MAP2K and MAPK activation / Formation of the canonical BAF (cBAF) complex / Formation of the polybromo-BAF (pBAF) complex / Formation of the embryonic stem cell BAF (esBAF) complex / Formation of the non-canonical BAF (ncBAF) complex / UCH proteinases / Gap junction degradation / Formation of annular gap junctions / RHOF GTPase cycle / Clathrin-mediated endocytosis / Regulation of CDH1 Function / Formation of the dystrophin-glycoprotein complex (DGC) / spectrin-associated cytoskeleton / negative regulation of substrate adhesion-dependent cell spreading / smooth endoplasmic reticulum calcium ion homeostasis / myofibril assembly / positive regulation of cellular component organization / platelet dense tubular network membrane / regulation of filopodium assembly / cell projection membrane / negative regulation of focal adhesion assembly / cellular response to cytochalasin B / regulation of transepithelial transport / morphogenesis of a polarized epithelium / structural constituent of postsynaptic actin cytoskeleton / COP9 signalosome / protein localization to adherens junction / dense body / Tat protein binding / regulation of lamellipodium assembly / postsynaptic actin cytoskeleton / actin filament capping / adherens junction assembly / apical protein localization / positive regulation of fibroblast migration / RHO GTPases activate IQGAPs / RHO GTPases Activate Formins / tight junction / ankyrin binding / apical junction complex / spectrin binding / positive regulation of wound healing / tropomyosin binding / regulation of norepinephrine uptake / transporter regulator activity / myofibril / NuA4 histone acetyltransferase complex / smooth endoplasmic reticulum / establishment or maintenance of cell polarity / cortical cytoskeleton / nitric-oxide synthase binding / brush border / striated muscle thin filament / regulation of synaptic vesicle endocytosis / kinesin binding / regulation of protein localization to plasma membrane / positive regulation of double-strand break repair via homologous recombination / erythrocyte development / cytoskeleton organization / axonogenesis / muscle contraction / cellular response to cAMP / calyx of Held / nitric-oxide synthase regulator activity / actin filament organization / adult locomotory behavior / actin filament / adherens junction / cell motility / SH3 domain binding / synapse organization / structural constituent of cytoskeleton / 加水分解酵素; 酸無水物に作用; 酸無水物に作用・細胞または細胞小器官の運動に関与 / Schaffer collateral - CA1 synapse / cytoplasmic ribonucleoprotein granule / actin filament binding / cell junction / nucleosome / regulation of cell shape / lamellipodium / actin cytoskeleton / actin binding / actin cytoskeleton organization / protein-containing complex assembly / cytoplasmic vesicle / cytoskeleton 類似検索 - 分子機能 | |||||||||
| 生物種 |  | |||||||||
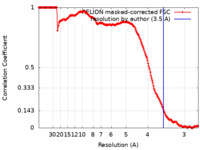
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.5 Å | |||||||||
 データ登録者 データ登録者 | Li N / Chen S / Gao N | |||||||||
| 資金援助 |  中国, 1件 中国, 1件
| |||||||||
 引用 引用 |  ジャーナル: Cell / 年: 2023 ジャーナル: Cell / 年: 2023タイトル: Structural basis of membrane skeleton organization in red blood cells. 著者: Ningning Li / Siyi Chen / Kui Xu / Meng-Ting He / Meng-Qiu Dong / Qiangfeng Cliff Zhang / Ning Gao /  要旨: The spectrin-based membrane skeleton is a ubiquitous membrane-associated two-dimensional cytoskeleton underneath the lipid membrane of metazoan cells. Mutations of skeleton proteins impair the ...The spectrin-based membrane skeleton is a ubiquitous membrane-associated two-dimensional cytoskeleton underneath the lipid membrane of metazoan cells. Mutations of skeleton proteins impair the mechanical strength and functions of the membrane, leading to several different types of human diseases. Here, we report the cryo-EM structures of the native spectrin-actin junctional complex (from porcine erythrocytes), which is a specialized short F-actin acting as the central organizational unit of the membrane skeleton. While an α-/β-adducin hetero-tetramer binds to the barbed end of F-actin as a flexible cap, tropomodulin and SH3BGRL2 together create an absolute cap at the pointed end. The junctional complex is strengthened by ring-like structures of dematin in the middle actin layers and by patterned periodic interactions with tropomyosin over its entire length. This work serves as a structural framework for understanding the assembly and dynamics of membrane skeleton and offers insights into mechanisms of various ubiquitous F-actin-binding factors in other F-actin systems. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_35302.map.gz emd_35302.map.gz | 398.9 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-35302-v30.xml emd-35302-v30.xml emd-35302.xml emd-35302.xml | 28.2 KB 28.2 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_35302_fsc.xml emd_35302_fsc.xml | 16.9 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_35302.png emd_35302.png | 29.7 KB | ||
| Filedesc metadata |  emd-35302.cif.gz emd-35302.cif.gz | 9.4 KB | ||
| その他 |  emd_35302_half_map_1.map.gz emd_35302_half_map_1.map.gz emd_35302_half_map_2.map.gz emd_35302_half_map_2.map.gz | 391.5 MB 391.5 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35302 http://ftp.pdbj.org/pub/emdb/structures/EMD-35302 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35302 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35302 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  8iaiMC  8iahC  8ib2C M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_35302.map.gz / 形式: CCP4 / 大きさ: 421.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_35302.map.gz / 形式: CCP4 / 大きさ: 421.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.37 Å | ||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: #2
| ファイル | emd_35302_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #1
| ファイル | emd_35302_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
+全体 : Spectrin-actin junctional complex
+超分子 #1: Spectrin-actin junctional complex
+分子 #1: Adducin 1
+分子 #2: Beta-adducin
+分子 #3: Dematin actin binding protein
+分子 #4: Actin, cytoplasmic 1
+分子 #5: Spectrin beta chain
+分子 #6: Tropomyosin-1.9
+分子 #7: Tropomyosin 3
+分子 #8: Tropomodulin-1
+分子 #9: SH3 domain-binding glutamic acid-rich-like protein
+分子 #10: ADENOSINE-5'-DIPHOSPHATE
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.5 |
|---|---|
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 QUANTUM (4k x 4k) 検出モード: SUPER-RESOLUTION / 平均電子線量: 34.4 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 3.0 µm / 最小 デフォーカス(公称値): 2.0 µm |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー

























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)