+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM map of nucleotide-free ABCA3 in LMNG/CHS | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 6.3 Å | |||||||||
 Authors Authors | Xie T / Zhang ZK / Yue J / Gong X | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Sci Adv / Year: 2022 Journal: Sci Adv / Year: 2022Title: Cryo-EM structures of the human surfactant lipid transporter ABCA3. Authors: Tian Xie / Zike Zhang / Jian Yue / Qi Fang / Xin Gong /  Abstract: The adenosine 5'-triphosphate (ATP)-binding cassette (ABC) transporter ABCA3 plays a critical role in pulmonary surfactant biogenesis. Mutations in human ABCA3 have been recognized as the most ...The adenosine 5'-triphosphate (ATP)-binding cassette (ABC) transporter ABCA3 plays a critical role in pulmonary surfactant biogenesis. Mutations in human ABCA3 have been recognized as the most frequent causes of inherited surfactant dysfunction disorders. Despite two decades of research, in vitro biochemical and structural studies of ABCA3 are still lacking. Here, we report the cryo-EM structures of human ABCA3 in two distinct conformations, both at resolution of 3.3 Å. In the absence of ATP, ABCA3 adopts a "lateral-opening" conformation with the lateral surfaces of transmembrane domains (TMDs) exposed to the membrane and features two positively charged cavities within the TMDs as potential substrate binding sites. ATP binding induces pronounced conformational changes, resulting in the collapse of the potential substrate binding cavities. Our results help to rationalize the disease-causing mutations in human ABCA3 and suggest a conserved "lateral access and extrusion" mechanism for both lipid export and import mediated by ABCA transporters. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_32235.map.gz emd_32235.map.gz | 49.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-32235-v30.xml emd-32235-v30.xml emd-32235.xml emd-32235.xml | 7.8 KB 7.8 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_32235.png emd_32235.png | 25.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-32235 http://ftp.pdbj.org/pub/emdb/structures/EMD-32235 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32235 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32235 | HTTPS FTP |
-Validation report
| Summary document |  emd_32235_validation.pdf.gz emd_32235_validation.pdf.gz | 341.8 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_32235_full_validation.pdf.gz emd_32235_full_validation.pdf.gz | 341.4 KB | Display | |
| Data in XML |  emd_32235_validation.xml.gz emd_32235_validation.xml.gz | 5.9 KB | Display | |
| Data in CIF |  emd_32235_validation.cif.gz emd_32235_validation.cif.gz | 6.7 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32235 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32235 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32235 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-32235 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_32235.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_32235.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : ABCA3
| Entire | Name: ABCA3 |
|---|---|
| Components |
|
-Supramolecule #1: ABCA3
| Supramolecule | Name: ABCA3 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
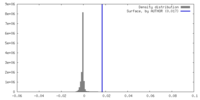
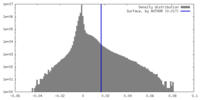
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 6.3 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 153740 |
|---|---|
| Initial angle assignment | Type: NOT APPLICABLE |
| Final angle assignment | Type: NOT APPLICABLE |
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)