+ Open data
Open data
- Basic information
Basic information
| Entry | 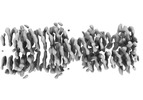 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | C4HR1_8r_shift5: Extendable repeat protein fiber | |||||||||
 Map data Map data | Sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Oligomer / repeat protein / DE NOVO PROTEIN | |||||||||
| Biological species | unidentified (others) | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 7.62 Å | |||||||||
 Authors Authors | Bethel NP / Borst AJ | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Chem / Year: 2023 Journal: Nat Chem / Year: 2023Title: Precisely patterned nanofibres made from extendable protein multiplexes. Authors: Neville P Bethel / Andrew J Borst / Fabio Parmeggiani / Matthew J Bick / T J Brunette / Hannah Nguyen / Alex Kang / Asim K Bera / Lauren Carter / Marcos C Miranda / Ryan D Kibler / Mila Lamb ...Authors: Neville P Bethel / Andrew J Borst / Fabio Parmeggiani / Matthew J Bick / T J Brunette / Hannah Nguyen / Alex Kang / Asim K Bera / Lauren Carter / Marcos C Miranda / Ryan D Kibler / Mila Lamb / Xinting Li / Banumathi Sankaran / David Baker /   Abstract: Molecular systems with coincident cyclic and superhelical symmetry axes have considerable advantages for materials design as they can be readily lengthened or shortened by changing the length of the ...Molecular systems with coincident cyclic and superhelical symmetry axes have considerable advantages for materials design as they can be readily lengthened or shortened by changing the length of the constituent monomers. Among proteins, alpha-helical coiled coils have such symmetric, extendable architectures, but are limited by the relatively fixed geometry and flexibility of the helical protomers. Here we describe a systematic approach to generating modular and rigid repeat protein oligomers with coincident C to C and superhelical symmetry axes that can be readily extended by repeat propagation. From these building blocks, we demonstrate that a wide range of unbounded fibres can be systematically designed by introducing hydrophilic surface patches that force staggering of the monomers; the geometry of such fibres can be precisely tuned by varying the number of repeat units in the monomer and the placement of the hydrophilic patches. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_29851.map.gz emd_29851.map.gz | 10.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-29851-v30.xml emd-29851-v30.xml emd-29851.xml emd-29851.xml | 16.5 KB 16.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_29851_fsc.xml emd_29851_fsc.xml | 8.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_29851.png emd_29851.png | 56.5 KB | ||
| Filedesc metadata |  emd-29851.cif.gz emd-29851.cif.gz | 4.8 KB | ||
| Others |  emd_29851_additional_1.map.gz emd_29851_additional_1.map.gz emd_29851_half_map_1.map.gz emd_29851_half_map_1.map.gz emd_29851_half_map_2.map.gz emd_29851_half_map_2.map.gz | 10.6 MB 59.2 MB 59.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-29851 http://ftp.pdbj.org/pub/emdb/structures/EMD-29851 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29851 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29851 | HTTPS FTP |
-Validation report
| Summary document |  emd_29851_validation.pdf.gz emd_29851_validation.pdf.gz | 1.1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_29851_full_validation.pdf.gz emd_29851_full_validation.pdf.gz | 1.1 MB | Display | |
| Data in XML |  emd_29851_validation.xml.gz emd_29851_validation.xml.gz | 16.6 KB | Display | |
| Data in CIF |  emd_29851_validation.cif.gz emd_29851_validation.cif.gz | 21.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29851 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29851 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29851 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29851 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_29851.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_29851.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened map | ||||||||||||||||||||
| Voxel size | X=Y=Z: 0.885 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Unsharpened map
| File | emd_29851_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unsharpened map | ||||||||||||
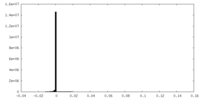
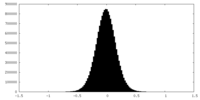
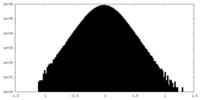
| Projections & Slices |
| ||||||||||||
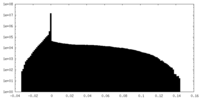
| Density Histograms |
-Half map: Half map A
| File | emd_29851_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
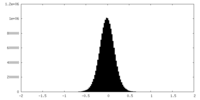
| Density Histograms |
-Half map: Half map B
| File | emd_29851_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
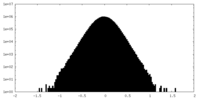
| Density Histograms |
- Sample components
Sample components
-Entire : C4HR1_8r_shift5
| Entire | Name: C4HR1_8r_shift5 |
|---|---|
| Components |
|
-Supramolecule #1: C4HR1_8r_shift5
| Supramolecule | Name: C4HR1_8r_shift5 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism: unidentified (others) |
| Molecular weight | Theoretical: 42.54 KDa |
-Macromolecule #1: C4HR1_8r_shift5
| Macromolecule | Name: C4HR1_8r_shift5 / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: unidentified (others) |
| Recombinant expression | Organism:  |
| Sequence | String: MMRDELIRWH LMHPMEQLRF LRELVENPER FREFWRRLEE EPGAVELFLE NIHLLHPIVQ IYLLELLVEN PELFRLFFEV LERCPGAVEL FLENWRLLPP IVQRIFLRLL VENPELFRLF FEVLERCPGA VKLFLENIKN LPPLIQKYLL RLLVENPELF RLFFEMLERC ...String: MMRDELIRWH LMHPMEQLRF LRELVENPER FREFWRRLEE EPGAVELFLE NIHLLHPIVQ IYLLELLVEN PELFRLFFEV LERCPGAVEL FLENWRLLPP IVQRIFLRLL VENPELFRLF FEVLERCPGA VKLFLENIKN LPPLIQKYLL RLLVENPELF RLFFEMLERC PGAVELFLEI IHELPPEIQK YFLELLVENP ELFRLFFEVL ERCPGAVKLF LNNIYLLHPL VQIHLLRLLV ENPELFRLFF EMLERCPGAV EQFLINLYLL HPIVQLVFLE LLVENPELFR LFFEVLKRCP GALELFKFII ELLPPIVQRH FKRLLEENPE LKELVKEVEE EGSLEHHHHH H |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | helical array |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS GLACIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.7000000000000001 µm |
 Movie
Movie Controller
Controller
















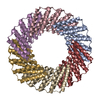

 Z
Z Y
Y X
X