[English] 日本語
 Yorodumi
Yorodumi- EMDB-28451: Cryo-EM Structure of Nanodisc reconstituted human ABCA7 EQ mutant... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM Structure of Nanodisc reconstituted human ABCA7 EQ mutant in ATP bound closed state | |||||||||
 Map data Map data | human ABCA7 EQ mutant in BPL/Ch nanodiscs | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationplasma membrane raft organization / apolipoprotein A-I receptor activity / positive regulation of engulfment of apoptotic cell / negative regulation of amyloid precursor protein biosynthetic process / phospholipid transporter activity / ABC transporters in lipid homeostasis / floppase activity / phosphatidylserine floppase activity / amyloid-beta clearance by cellular catabolic process / positive regulation of phospholipid efflux ...plasma membrane raft organization / apolipoprotein A-I receptor activity / positive regulation of engulfment of apoptotic cell / negative regulation of amyloid precursor protein biosynthetic process / phospholipid transporter activity / ABC transporters in lipid homeostasis / floppase activity / phosphatidylserine floppase activity / amyloid-beta clearance by cellular catabolic process / positive regulation of phospholipid efflux / negative regulation of endocytosis / phosphatidylcholine floppase activity / peptide cross-linking / phospholipid efflux / high-density lipoprotein particle assembly / positive regulation of amyloid-beta clearance / P-type phospholipid transporter / positive regulation of protein localization to cell surface / apolipoprotein A-I-mediated signaling pathway / regulation of amyloid precursor protein catabolic process / phospholipid translocation / cholesterol efflux / negative regulation of PERK-mediated unfolded protein response / negative regulation of amyloid-beta formation / amyloid-beta formation / glial cell projection / ATPase-coupled transmembrane transporter activity / phagocytic cup / regulation of lipid metabolic process / protein localization to nucleus / ABC-type transporter activity / positive regulation of cholesterol efflux / negative regulation of MAPK cascade / phagocytosis / positive regulation of phagocytosis / visual learning / memory / ruffle membrane / cell junction / early endosome membrane / positive regulation of ERK1 and ERK2 cascade / Golgi membrane / intracellular membrane-bounded organelle / Golgi apparatus / cell surface / endoplasmic reticulum / ATP hydrolysis activity / ATP binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.7 Å | |||||||||
 Authors Authors | Alam A / Le LTM / Thompson JR | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: EMBO J / Year: 2023 Journal: EMBO J / Year: 2023Title: Cryo-EM structures of human ABCA7 provide insights into its phospholipid translocation mechanisms. Authors: Le Thi My Le / James Robert Thompson / Sepehr Dehghani-Ghahnaviyeh / Shashank Pant / Phuoc Xuan Dang / Jarrod Bradley French / Takahisa Kanikeyo / Emad Tajkhorshid / Amer Alam /  Abstract: Phospholipid extrusion by ABC subfamily A (ABCA) exporters is central to cellular physiology, although the specifics of the underlying substrate interactions and transport mechanisms remain poorly ...Phospholipid extrusion by ABC subfamily A (ABCA) exporters is central to cellular physiology, although the specifics of the underlying substrate interactions and transport mechanisms remain poorly resolved at the molecular level. Here we report cryo-EM structures of lipid-embedded human ABCA7 in an open state and in a nucleotide-bound, closed state at resolutions between 3.6 and 4.0 Å. The former reveals an ordered patch of bilayer lipids traversing the transmembrane domain (TMD), while the latter reveals a lipid-free, closed TMD with a small extracellular opening. These structures offer a structural framework for both substrate entry and exit from the ABCA7 TMD and highlight conserved rigid-body motions that underlie the associated conformational transitions. Combined with functional analysis and molecular dynamics (MD) simulations, our data also shed light on lipid partitioning into the ABCA7 TMD and localized membrane perturbations that underlie ABCA7 function and have broader implications for other ABCA family transporters. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_28451.map.gz emd_28451.map.gz | 202.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-28451-v30.xml emd-28451-v30.xml emd-28451.xml emd-28451.xml | 21.8 KB 21.8 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_28451.png emd_28451.png | 20.5 KB | ||
| Others |  emd_28451_additional_1.map.gz emd_28451_additional_1.map.gz emd_28451_half_map_1.map.gz emd_28451_half_map_1.map.gz emd_28451_half_map_2.map.gz emd_28451_half_map_2.map.gz | 128.3 MB 171.7 MB 171.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-28451 http://ftp.pdbj.org/pub/emdb/structures/EMD-28451 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28451 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28451 | HTTPS FTP |
-Validation report
| Summary document |  emd_28451_validation.pdf.gz emd_28451_validation.pdf.gz | 779.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_28451_full_validation.pdf.gz emd_28451_full_validation.pdf.gz | 779.1 KB | Display | |
| Data in XML |  emd_28451_validation.xml.gz emd_28451_validation.xml.gz | 15.3 KB | Display | |
| Data in CIF |  emd_28451_validation.cif.gz emd_28451_validation.cif.gz | 18.1 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28451 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28451 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28451 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28451 | HTTPS FTP |
-Related structure data
| Related structure data |  8eopMC  8edwC  8ee6C  8eebC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_28451.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_28451.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | human ABCA7 EQ mutant in BPL/Ch nanodiscs | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.889 Å | ||||||||||||||||||||||||||||||||||||
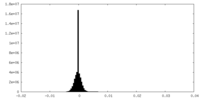
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Local resolution filtered map
| File | emd_28451_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Local resolution filtered map | ||||||||||||
| Projections & Slices |
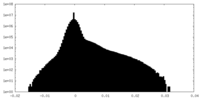
| ||||||||||||
| Density Histograms |
-Half map: Refinement half 1 map
| File | emd_28451_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Refinement half 1 map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Refinement half 2 map
| File | emd_28451_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Refinement half 2 map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Human ABCA7 eq mutant in Porcine BPL/Ch/MSP1D1 nanodiscs in compl...
| Entire | Name: Human ABCA7 eq mutant in Porcine BPL/Ch/MSP1D1 nanodiscs in complex with ATP//Mg2+ |
|---|---|
| Components |
|
-Supramolecule #1: Human ABCA7 eq mutant in Porcine BPL/Ch/MSP1D1 nanodiscs in compl...
| Supramolecule | Name: Human ABCA7 eq mutant in Porcine BPL/Ch/MSP1D1 nanodiscs in complex with ATP//Mg2+ type: complex / ID: 1 / Chimera: Yes / Parent: 0 / Macromolecule list: #1 Details: Human ABCA7 EQ mutant recombinantly expressed in HEK293 TREX cell line and reconstituted in MSP1D1 nanodiscs comprising 4:1 mixture of Porcine Brain Polar Lipids:Cholesterol |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 234.35 KDa |
-Macromolecule #1: Phospholipid-transporting ATPase ABCA7
| Macromolecule | Name: Phospholipid-transporting ATPase ABCA7 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO / EC number: P-type phospholipid transporter |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 234.596609 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MAFWTQLMLL LWKNFMYRRR QPVQLLVELL WPLFLFFILV AVRHSHPPLE HHECHFPNKP LPSAGTVPWL QGLICNVNNT CFPQLTPGE EPGRLSNFND SLVSRLLADA RTVLGGASAH RTLAGLGKLI ATLRAARSTA QPQPTKQSPL EPPMLDVAEL L TSLLRTES ...String: MAFWTQLMLL LWKNFMYRRR QPVQLLVELL WPLFLFFILV AVRHSHPPLE HHECHFPNKP LPSAGTVPWL QGLICNVNNT CFPQLTPGE EPGRLSNFND SLVSRLLADA RTVLGGASAH RTLAGLGKLI ATLRAARSTA QPQPTKQSPL EPPMLDVAEL L TSLLRTES LGLALGQAQE PLHSLLEAAE DLAQELLALR SLVELRALLQ RPRGTSGPLE LLSEALCSVR GPSSTVGPSL NW YEASDLM ELVGQEPESA LPDSSLSPAC SELIGALDSH PLSRLLWRRL KPLILGKLLF APDTPFTRKL MAQVNRTFEE LTL LRDVRE VWEMLGPRIF TFMNDSSNVA MLQRLLQMQD EGRRQPRPGG RDHMEALRSF LDPGSGGYSW QDAHADVGHL VGTL GRVTE CLSLDKLEAA PSEAALVSRA LQLLAEHRFW AGVVFLGPED SSDPTEHPTP DLGPGHVRIK IRMDIDVVTR TNKIR DRFW DPGPAADPLT DLRYVWGGFV YLQDLVERAA VRVLSGANPR AGLYLQQMPY PCYVDDVFLR VLSRSLPLFL TLAWIY SVT LTVKAVVREK ETRLRDTMRA MGLSRAVLWL GWFLSCLGPF LLSAALLVLV LKLGDILPYS HPGVVFLFLA AFAVATV TQ SFLLSAFFSR ANLAAACGGL AYFSLYLPYV LCVAWRDRLP AGGRVAASLL SPVAFGFGCE SLALLEEQGE GAQWHNVG T RPTADVFSLA QVSGLLLLDA ALYGLATWYL EAVCPGQYGI PEPWNFPFRR SYWCGPRPPK SPAPCPTPLD PKVLVEEAP PGLSPGVSVR SLEKRFPGSP QPALRGLSLD FYQGHITAFL GHNGAGKTTT LSILSGLFPP SGGSAFILGH DVRSSMAAIR PHLGVCPQY NVLFDMLTVD EHVWFYGRLK GLSAAVVGPE QDRLLQDVGL VSKQSVQTRH LSGGMQRKLS VAIAFVGGSQ V VILDQPTA GVDPASRRGI WELLLKYREG RTLILSTHHL DEAELLGDRV AVVAGGRLCC CGSPLFLRRH LGSGYYLTLV KA RLPLTTN EKADTDMEGS VDTRQEKKNG SQGSRVGTPQ LLALVQHWVP GARLVEELPH ELVLVLPYTG AHDGSFATLF REL DTRLAE LRLTGYGISD TSLEEIFLKV VEECAADTDM EDGSCGQHLC TGIAGLDVTL RLKMPPQETA LENGEPAGSA PETD QGSGP DAVGRVQGWA LTRQQLQALL LKRFLLARRS RRGLFAQIVL PALFVGLALV FSLIVPPFGH YPALRLSPTM YGAQV SFFS EDAPGDPGRA RLLEALLQEA GLEEPPVQHS SHRFSAPEVP AEVAKVLASG NWTPESPSPA CQCSRPGARR LLPDCP AAA GGPPPPQAVT GSGEVVQNLT GRNLSDFLVK TYPRLVRQGL KTKKWVNEVR YGGFSLGGRD PGLPSGQELG RSVEELW AL LSPLPGGALD RVLKNLTAWA HSLDAQDSLK IWFNNKGWHS MVAFVNRASN AILRAHLPPG PARHAHSITT LNHPLNLT K EQLSEGALMA SSVDVLVSIC VVFAMSFVPA SFTLVLIEER VTRAKHLQLM GGLSPTLYWL GNFLWDMCNY LVPACIVVL IFLAFQQRAY VAPANLPALL LLLLLYGWSI TPLMYPASFF FSVPSTAYVV LTCINLFIGI NGSMATFVLE LFSDQKLQEV SRILKQVFL IFPHFCLGRG LIDMVRNQAM ADAFERLGDR QFQSPLRWEV VGKNLLAMVI QGPLFLLFTL LLQHRSQLLP Q PRVRSLPL LGEEDEDVAR ERERVVQGAT QGDVLVLRNL TKVYRGQRMP AVDRLCLGIP PGECFGLLGV NGAGKTSTFR MV TGDTLAS RGEAVLAGHS VAREPSAAHL SMGYCPQSDA IFELLTGREH LELLARLRGV PEAQVAQTAG SGLARLGLSW YAD RPAGTY SGGNKRKLAT ALALVGDPAV VFLDQPTTGM DPSARRFLWN SLLAVVREGR SVMLTSHSME ECEALCSRLA IMVN GRFRC LGSPQHLKGR FAAGHTLTLR VPAARSQPAA AFVAAEFPGA ELREAHGGRL RFQLPPGGRC ALARVFGELA VHGAE HGVE DFSVSQTMLE EVFLYFSKDQ GKDEDTEEQK EAGVGVDPAP GLQHPKRVSQ FLDDPSTAET VL |
-Macromolecule #4: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 4 / Number of copies: 6 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Macromolecule #5: ADENOSINE-5'-TRIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-TRIPHOSPHATE / type: ligand / ID: 5 / Number of copies: 2 / Formula: ATP |
|---|---|
| Molecular weight | Theoretical: 507.181 Da |
| Chemical component information |  ChemComp-ATP: |
-Macromolecule #6: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 6 / Number of copies: 2 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.5 mg/mL |
|---|---|
| Buffer | pH: 7.5 Details: 25mM HEPES pH 7.5, 150mM NaCl, 5mM ATP, 10mM Magnesium Chloride |
| Grid | Model: Quantifoil R1.2/1.3 / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: COUNTING / Average exposure time: 60.0 sec. / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.4 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)