[English] 日本語
 Yorodumi
Yorodumi- EMDB-2417: Negative staining 3D-EM of the progenitor toxin complex (PTC) of ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-2417 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Negative staining 3D-EM of the progenitor toxin complex (PTC) of Botulinum toxin type A | |||||||||
 Map data Map data | negative staining 3D-EM of the the progenitor toxin complex (PTC) of Botulinum toxin type A | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Botulinum toxin / the progenitor toxin complex (PTC) | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / negative staining / Resolution: 30.0 Å | |||||||||
 Authors Authors | Lee K / Gu S / Jin L / Le TTN / Cheng LW / Strotmeier J / Kruel AM / Yao G / Perry K / Rummel A / Jin R | |||||||||
 Citation Citation |  Journal: PLoS Pathog / Year: 2013 Journal: PLoS Pathog / Year: 2013Title: Structure of a bimodular botulinum neurotoxin complex provides insights into its oral toxicity. Authors: Kwangkook Lee / Shenyan Gu / Lei Jin / Thi Tuc Nghi Le / Luisa W Cheng / Jasmin Strotmeier / Anna Magdalena Kruel / Guorui Yao / Kay Perry / Andreas Rummel / Rongsheng Jin /  Abstract: Botulinum neurotoxins (BoNTs) are produced by Clostridium botulinum and cause the fatal disease botulism, a flaccid paralysis of the muscle. BoNTs are released together with several auxiliary ...Botulinum neurotoxins (BoNTs) are produced by Clostridium botulinum and cause the fatal disease botulism, a flaccid paralysis of the muscle. BoNTs are released together with several auxiliary proteins as progenitor toxin complexes (PTCs) to become highly potent oral poisons. Here, we report the structure of a ∼760 kDa 14-subunit large PTC of serotype A (L-PTC/A) and reveal insight into its absorption mechanism. Using a combination of X-ray crystallography, electron microscopy, and functional studies, we found that L-PTC/A consists of two structurally and functionally independent sub-complexes. A hetero-dimeric 290 kDa complex protects BoNT, while a hetero-dodecameric 470 kDa complex facilitates its absorption in the harsh environment of the gastrointestinal tract. BoNT absorption is mediated by nine glycan-binding sites on the dodecameric sub-complex that forms multivalent interactions with carbohydrate receptors on intestinal epithelial cells. We identified monosaccharides that blocked oral BoNT intoxication in mice, which suggests a new strategy for the development of preventive countermeasures for BoNTs based on carbohydrate receptor mimicry. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_2417.map.gz emd_2417.map.gz | 2.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-2417-v30.xml emd-2417-v30.xml emd-2417.xml emd-2417.xml | 12.6 KB 12.6 KB | Display Display |  EMDB header EMDB header |
| Images |  EMD-2417_500x500.jpg EMD-2417_500x500.jpg | 117.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2417 http://ftp.pdbj.org/pub/emdb/structures/EMD-2417 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2417 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2417 | HTTPS FTP |
-Validation report
| Summary document |  emd_2417_validation.pdf.gz emd_2417_validation.pdf.gz | 181.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_2417_full_validation.pdf.gz emd_2417_full_validation.pdf.gz | 180.5 KB | Display | |
| Data in XML |  emd_2417_validation.xml.gz emd_2417_validation.xml.gz | 5.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2417 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2417 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2417 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2417 | HTTPS FTP |
-Related structure data
| Related structure data | 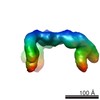 2416C  4lo0C  4lo1C  4lo2C  4lo3C  4lo4C  4lo5C  4lo6C  4lo7C  4lo8C C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_2417.map.gz / Format: CCP4 / Size: 5.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_2417.map.gz / Format: CCP4 / Size: 5.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | negative staining 3D-EM of the the progenitor toxin complex (PTC) of Botulinum toxin type A | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 4.28 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : The progenitor toxin complexes of Botulinum toxin type A
| Entire | Name: The progenitor toxin complexes of Botulinum toxin type A |
|---|---|
| Components |
|
-Supramolecule #1000: The progenitor toxin complexes of Botulinum toxin type A
| Supramolecule | Name: The progenitor toxin complexes of Botulinum toxin type A type: sample / ID: 1000 Details: The complex is monodisperse, and stable only at acidic pH. Oligomeric state: A 14-subunit complex of 1 BoNT/A, 1 NTNHA, 3 HA70, 3 HA17 and 6 HA33 Number unique components: 5 |
|---|---|
| Molecular weight | Experimental: 760 KDa / Theoretical: 760 KDa |
-Macromolecule #1: BoNT/A
| Macromolecule | Name: BoNT/A / type: protein_or_peptide / ID: 1 / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
-Macromolecule #2: NTNHA
| Macromolecule | Name: NTNHA / type: protein_or_peptide / ID: 2 / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 760 KDa |
| Recombinant expression | Organism:  |
-Macromolecule #3: HA17
| Macromolecule | Name: HA17 / type: protein_or_peptide / ID: 3 / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
-Macromolecule #4: HA33
| Macromolecule | Name: HA33 / type: protein_or_peptide / ID: 4 / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
-Macromolecule #5: HA70
| Macromolecule | Name: HA70 / type: protein_or_peptide / ID: 5 / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
-Experimental details
-Structure determination
| Method | negative staining |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.02 mg/mL |
|---|---|
| Buffer | pH: 6.2 / Details: 100mM NaCl, 20mM MES |
| Staining | Type: NEGATIVE Details: Grids with adsorbed protein floated on 1% w/v uranyl formate for 30 seconds |
| Grid | Details: 300 mesh copper grid with carbon support, glow discharged in vacuumed air |
| Vitrification | Cryogen name: NONE / Instrument: OTHER |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F20 |
|---|---|
| Alignment procedure | Legacy - Astigmatism: Objective lens astigmatism was corrected at 70,000 times magnification. |
| Date | Feb 4, 2012 |
| Image recording | Category: CCD / Film or detector model: GENERIC TVIPS (4k x 4k) / Number real images: 362 / Average electron dose: 40 e/Å2 / Bits/pixel: 8 |
| Tilt angle min | 0 |
| Tilt angle max | 0 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 70000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 3.6 µm / Nominal defocus min: 1.5 µm |
| Sample stage | Specimen holder model: SIDE ENTRY, EUCENTRIC |
| Experimental equipment |  Model: Tecnai F20 / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | Image processing was done using EMAN2.The particles were selected by a semi-automatic selection using e2boxer. |
|---|---|
| CTF correction | Details: Each particle |
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 30.0 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: EMAN2 / Number images used: 15140 |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)