+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-2354 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Anaphase Promoting Complex with full length Emi1 bound | |||||||||
 Map data Map data | Anaphase Promoting Complex - Emi1 (full length) | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | cell cycle regulation | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / negative staining / Resolution: 20.0 Å | |||||||||
 Authors Authors | Frye JJ / Brown NG / Petzold G / Watson ER / Grace CRR / Nourse A / Jarvis M / Kriwacki RW / Peters JM / Stark H / Schulman BA | |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2013 Journal: Nat Struct Mol Biol / Year: 2013Title: Electron microscopy structure of human APC/C(CDH1)-EMI1 reveals multimodal mechanism of E3 ligase shutdown. Authors: Jeremiah J Frye / Nicholas G Brown / Georg Petzold / Edmond R Watson / Christy R R Grace / Amanda Nourse / Marc A Jarvis / Richard W Kriwacki / Jan-Michael Peters / Holger Stark / Brenda A Schulman /  Abstract: The anaphase-promoting complex/cyclosome (APC/C) is a ~1.5-MDa multiprotein E3 ligase enzyme that regulates cell division by promoting timely ubiquitin-mediated proteolysis of key cell-cycle ...The anaphase-promoting complex/cyclosome (APC/C) is a ~1.5-MDa multiprotein E3 ligase enzyme that regulates cell division by promoting timely ubiquitin-mediated proteolysis of key cell-cycle regulatory proteins. Inhibition of human APC/C(CDH1) during interphase by early mitotic inhibitor 1 (EMI1) is essential for accurate coordination of DNA synthesis and mitosis. Here, we report a hybrid structural approach involving NMR, electron microscopy and enzymology, which reveal that EMI1's 143-residue C-terminal domain inhibits multiple APC/C(CDH1) functions. The intrinsically disordered D-box, linker and tail elements, together with a structured zinc-binding domain, bind distinct regions of APC/C(CDH1) to synergistically both block the substrate-binding site and inhibit ubiquitin-chain elongation. The functional importance of intrinsic structural disorder is explained by enabling a small inhibitory domain to bind multiple sites to shut down various functions of a 'molecular machine' nearly 100 times its size. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_2354.map.gz emd_2354.map.gz | 1.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-2354-v30.xml emd-2354-v30.xml emd-2354.xml emd-2354.xml | 8.3 KB 8.3 KB | Display Display |  EMDB header EMDB header |
| Images |  EMD-2354_full_length_view.jpg EMD-2354_full_length_view.jpg | 36.2 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2354 http://ftp.pdbj.org/pub/emdb/structures/EMD-2354 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2354 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2354 | HTTPS FTP |
-Validation report
| Summary document |  emd_2354_validation.pdf.gz emd_2354_validation.pdf.gz | 198.2 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_2354_full_validation.pdf.gz emd_2354_full_validation.pdf.gz | 197.4 KB | Display | |
| Data in XML |  emd_2354_validation.xml.gz emd_2354_validation.xml.gz | 4.7 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2354 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2354 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2354 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2354 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_2354.map.gz / Format: CCP4 / Size: 1.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_2354.map.gz / Format: CCP4 / Size: 1.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Anaphase Promoting Complex - Emi1 (full length) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 5 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
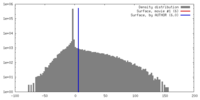
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : APC/C - Emi1 complex
| Entire | Name: APC/C - Emi1 complex |
|---|---|
| Components |
|
-Supramolecule #1000: APC/C - Emi1 complex
| Supramolecule | Name: APC/C - Emi1 complex / type: sample / ID: 1000 / Number unique components: 2 |
|---|---|
| Molecular weight | Theoretical: 1.6 MDa |
-Macromolecule #1: APC/C
| Macromolecule | Name: APC/C / type: protein_or_peptide / ID: 1 / Name.synonym: Anaphase Promoting Complex / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human Homo sapiens (human) / synonym: Human |
-Macromolecule #2: Emi1
| Macromolecule | Name: Emi1 / type: protein_or_peptide / ID: 2 / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human Homo sapiens (human) / synonym: Human |
-Experimental details
-Structure determination
| Method | negative staining |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 / Details: 20mM Tris-HCL, 150 mM NaCl |
|---|---|
| Staining | Type: NEGATIVE / Details: Uranyl Formate |
| Vitrification | Cryogen name: NONE / Instrument: OTHER |
- Electron microscopy
Electron microscopy
| Microscope | FEI/PHILIPS CM200FEG |
|---|---|
| Date | Jun 13, 2012 |
| Image recording | Category: CCD / Film or detector model: GENERIC CCD / Average electron dose: 25 e/Å2 |
| Electron beam | Acceleration voltage: 160 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 118800 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2 mm |
| Sample stage | Specimen holder model: SIDE ENTRY, EUCENTRIC |
- Image processing
Image processing
| CTF correction | Details: Each particle |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 20.0 Å / Software - Name: Imagic / Number images used: 18911 |
 Movie
Movie Controller
Controller













 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)