[English] 日本語
 Yorodumi
Yorodumi- EMDB-17072: Virus-like Particle based on PVY coat protein with T43C and D136C... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Virus-like Particle based on PVY coat protein with T43C and D136C mutation with helical architecture encapsidating ssRNA | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | helical / VLP / ssRNA / Potyvirus / PVY / VIRUS LIKE PARTICLE | |||||||||
| Function / homology | Potyvirus coat protein / Potyvirus coat protein / viral capsid / Genome polyprotein Function and homology information Function and homology information | |||||||||
| Biological species |  Potato virus Y strain NTN Potato virus Y strain NTN | |||||||||
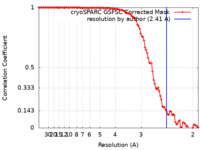
| Method | helical reconstruction / cryo EM / Resolution: 2.41 Å | |||||||||
 Authors Authors | Kavcic L / Kezar A / Podobnik M | |||||||||
| Funding support |  Slovenia, 2 items Slovenia, 2 items
| |||||||||
 Citation Citation |  Journal: Commun Chem / Year: 2024 Journal: Commun Chem / Year: 2024Title: From structural polymorphism to structural metamorphosis of the coat protein of flexuous filamentous potato virus Y. Authors: Luka Kavčič / Andreja Kežar / Neža Koritnik / Magda Tušek Žnidarič / Tajda Klobučar / Žiga Vičič / Franci Merzel / Ellie Holden / Justin L P Benesch / Marjetka Podobnik /   Abstract: The structural diversity and tunability of the capsid proteins (CPs) of various icosahedral and rod-shaped viruses have been well studied and exploited in the development of smart hybrid ...The structural diversity and tunability of the capsid proteins (CPs) of various icosahedral and rod-shaped viruses have been well studied and exploited in the development of smart hybrid nanoparticles. However, the potential of CPs of the wide-spread flexuous filamentous plant viruses remains to be explored. Here, we show that we can control the shape, size, RNA encapsidation ability, symmetry, stability and surface functionalization of nanoparticles through structure-based design of CP from potato virus Y (PVY). We provide high-resolution insight into CP-based self-assemblies, ranging from large polymorphic or monomorphic filaments to smaller annular, cubic or spherical particles. Furthermore, we show that we can prevent CP self-assembly in bacteria by fusion with a cleavable protein, enabling controlled nanoparticle formation in vitro. Understanding the remarkable structural diversity of PVY CP not only provides possibilities for the production of biodegradable nanoparticles, but may also advance future studies of CP's polymorphism in a biological context. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17072.map.gz emd_17072.map.gz | 23 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17072-v30.xml emd-17072-v30.xml emd-17072.xml emd-17072.xml | 18.4 KB 18.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_17072_fsc.xml emd_17072_fsc.xml | 9.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_17072.png emd_17072.png | 91.6 KB | ||
| Masks |  emd_17072_msk_1.map emd_17072_msk_1.map | 103 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-17072.cif.gz emd-17072.cif.gz | 6.1 KB | ||
| Others |  emd_17072_additional_1.map.gz emd_17072_additional_1.map.gz emd_17072_half_map_1.map.gz emd_17072_half_map_1.map.gz emd_17072_half_map_2.map.gz emd_17072_half_map_2.map.gz | 51.3 MB 95.3 MB 95.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17072 http://ftp.pdbj.org/pub/emdb/structures/EMD-17072 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17072 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17072 | HTTPS FTP |
-Related structure data
| Related structure data |  8oplMC  8opaC  8opbC  8opcC  8opdC  8opeC  8opfC  8opgC  8ophC  8opjC  8opkC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_17072.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17072.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.95 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_17072_msk_1.map emd_17072_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
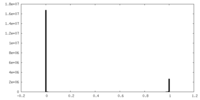
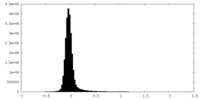
| Density Histograms |
-Additional map: #1
| File | emd_17072_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
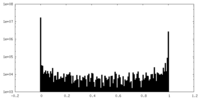
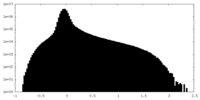
| Density Histograms |
-Half map: #2
| File | emd_17072_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
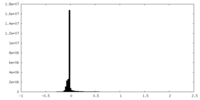
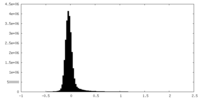
| Density Histograms |
-Half map: #1
| File | emd_17072_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
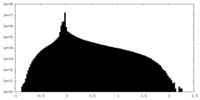
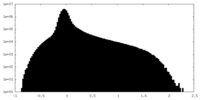
| Density Histograms |
- Sample components
Sample components
-Entire : Virus-like particles from PVY coat protein with T43C and D136C mu...
| Entire | Name: Virus-like particles from PVY coat protein with T43C and D136C mutations |
|---|---|
| Components |
|
-Supramolecule #1: Virus-like particles from PVY coat protein with T43C and D136C mu...
| Supramolecule | Name: Virus-like particles from PVY coat protein with T43C and D136C mutations type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: Monomorphic filaments with helical architecture and encapsidated RNA |
|---|---|
| Source (natural) | Organism:  Potato virus Y strain NTN Potato virus Y strain NTN |
-Macromolecule #1: Genome polyprotein (Fragment)
| Macromolecule | Name: Genome polyprotein (Fragment) / type: protein_or_peptide / ID: 1 / Number of copies: 27 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Potato virus Y strain NTN Potato virus Y strain NTN |
| Molecular weight | Theoretical: 29.925891 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GNDTIDAGGS TKKDAKQEQG SIQPNLNKEK EKDVNVGTSG THCVPRIKAI TSKMRMPKSK GATVLNLEHL LEYAPQQIDI SNTRATQSQ FDTWYEAVQL AYDIGETEMP TVMNGLMVWC IENGTSPNIN GVWVMMCGDE QVEYPLKPIV ENAKPTLRQI M AHFSDVAE ...String: GNDTIDAGGS TKKDAKQEQG SIQPNLNKEK EKDVNVGTSG THCVPRIKAI TSKMRMPKSK GATVLNLEHL LEYAPQQIDI SNTRATQSQ FDTWYEAVQL AYDIGETEMP TVMNGLMVWC IENGTSPNIN GVWVMMCGDE QVEYPLKPIV ENAKPTLRQI M AHFSDVAE AYIEMRNKKE PYMPRYGLVR NLRDGSLARY AFDFYEVTSR TPVRAREAHI QMKAAALKSA QSRLFGLDGG IS TQEENTE RHTTEDVSPS MHTLLGVKNM UniProtKB: Genome polyprotein |
-Macromolecule #2: RNA (5'-R(P*UP*UP*UP*UP*U)-3')
| Macromolecule | Name: RNA (5'-R(P*UP*UP*UP*UP*U)-3') / type: rna / ID: 2 / Number of copies: 27 |
|---|---|
| Source (natural) | Organism:  Potato virus Y strain NTN Potato virus Y strain NTN |
| Molecular weight | Theoretical: 1.485872 KDa |
| Sequence | String: UUUUU |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL |
|---|---|
| Buffer | pH: 7.4 Details: 1.8 mM KH2PO4, 10.1 mM Na2HPO4, 140 mM NaCl, 2.7 mM KCl, pH 7.4 |
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY ARRAY |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS GLACIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: COUNTING / Number real images: 461 / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.1 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 150000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
 Movie
Movie Controller
Controller
































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)