[English] 日本語
 Yorodumi
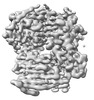
Yorodumi- EMDB-14881: Chaetomium thermophilum Mre11-Rad50-Nbs1 complex bound to ATPyS (... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Chaetomium thermophilum Mre11-Rad50-Nbs1 complex bound to ATPyS (composite structure) | ||||||||||||
 Map data Map data | Composite map | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | DNA repair / complex / ATPase / coiled-coils / HYDROLASE | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationmitochondrial double-strand break repair via homologous recombination / Mre11 complex / meiotic DNA double-strand break formation / chromosome organization involved in meiotic cell cycle / Hydrolases; Acting on acid anhydrides / G-quadruplex DNA binding / double-stranded telomeric DNA binding / single-stranded DNA endodeoxyribonuclease activity / telomere maintenance via recombination / single-stranded telomeric DNA binding ...mitochondrial double-strand break repair via homologous recombination / Mre11 complex / meiotic DNA double-strand break formation / chromosome organization involved in meiotic cell cycle / Hydrolases; Acting on acid anhydrides / G-quadruplex DNA binding / double-stranded telomeric DNA binding / single-stranded DNA endodeoxyribonuclease activity / telomere maintenance via recombination / single-stranded telomeric DNA binding / mitotic intra-S DNA damage checkpoint signaling / DNA duplex unwinding / 3'-5'-DNA exonuclease activity / mitotic G2 DNA damage checkpoint signaling / telomere maintenance via telomerase / telomere maintenance / guanyl-nucleotide exchange factor activity / condensed nuclear chromosome / double-strand break repair via homologous recombination / double-strand break repair via nonhomologous end joining / double-strand break repair / site of double-strand break / manganese ion binding / double-stranded DNA binding / Hydrolases; Acting on ester bonds / ATP hydrolysis activity / metal ion binding Similarity search - Function | ||||||||||||
| Biological species |  Chaetomium thermophilum (fungus) / Chaetomium thermophilum (fungus) /  Thermochaetoides thermophila (fungus) Thermochaetoides thermophila (fungus) | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.0 Å | ||||||||||||
 Authors Authors | Rotheneder M / Stakyte K / Bartho JD / Lammens K / Hopfner KP | ||||||||||||
| Funding support |  Germany, 3 items Germany, 3 items
| ||||||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2023 Journal: Mol Cell / Year: 2023Title: Cryo-EM structure of the Mre11-Rad50-Nbs1 complex reveals the molecular mechanism of scaffolding functions. Authors: Matthias Rotheneder / Kristina Stakyte / Erik van de Logt / Joseph D Bartho / Katja Lammens / Yilan Fan / Aaron Alt / Brigitte Kessler / Christophe Jung / Wynand P Roos / Barbara ...Authors: Matthias Rotheneder / Kristina Stakyte / Erik van de Logt / Joseph D Bartho / Katja Lammens / Yilan Fan / Aaron Alt / Brigitte Kessler / Christophe Jung / Wynand P Roos / Barbara Steigenberger / Karl-Peter Hopfner /  Abstract: The DNA double-strand break repair complex Mre11-Rad50-Nbs1 (MRN) detects and nucleolytically processes DNA ends, activates the ATM kinase, and tethers DNA at break sites. How MRN can act both as ...The DNA double-strand break repair complex Mre11-Rad50-Nbs1 (MRN) detects and nucleolytically processes DNA ends, activates the ATM kinase, and tethers DNA at break sites. How MRN can act both as nuclease and scaffold protein is not well understood. The cryo-EM structure of MRN from Chaetomium thermophilum reveals a 2:2:1 complex with a single Nbs1 wrapping around the autoinhibited Mre11 nuclease dimer. MRN has two DNA-binding modes, one ATP-dependent mode for loading onto DNA ends and one ATP-independent mode through Mre11's C terminus, suggesting how it may interact with DSBs and intact DNA. MRNs two 60-nm-long coiled-coil domains form a linear rod structure, the apex of which is assembled by the two joined zinc-hook motifs. Apices from two MRN complexes can further dimerize, forming 120-nm spanning MRN-MRN structures. Our results illustrate the architecture of MRN and suggest how it mechanistically integrates catalytic and tethering functions. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_14881.map.gz emd_14881.map.gz | 230 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-14881-v30.xml emd-14881-v30.xml emd-14881.xml emd-14881.xml | 19.1 KB 19.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_14881.png emd_14881.png | 30 KB | ||
| Filedesc metadata |  emd-14881.cif.gz emd-14881.cif.gz | 8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-14881 http://ftp.pdbj.org/pub/emdb/structures/EMD-14881 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14881 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14881 | HTTPS FTP |
-Validation report
| Summary document |  emd_14881_validation.pdf.gz emd_14881_validation.pdf.gz | 275.9 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_14881_full_validation.pdf.gz emd_14881_full_validation.pdf.gz | 275.5 KB | Display | |
| Data in XML |  emd_14881_validation.xml.gz emd_14881_validation.xml.gz | 6.6 KB | Display | |
| Data in CIF |  emd_14881_validation.cif.gz emd_14881_validation.cif.gz | 7.7 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14881 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14881 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14881 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14881 | HTTPS FTP |
-Related structure data
| Related structure data |  7zr1MC  7zqyC  8bahC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_14881.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_14881.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Composite map | ||||||||||||||||||||
| Voxel size | X=Y=Z: 2.03328 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : Chaetomium thermophilum Mre11-Rad50-Nbs1 complex bound to ATPyS
| Entire | Name: Chaetomium thermophilum Mre11-Rad50-Nbs1 complex bound to ATPyS |
|---|---|
| Components |
|
-Supramolecule #1: Chaetomium thermophilum Mre11-Rad50-Nbs1 complex bound to ATPyS
| Supramolecule | Name: Chaetomium thermophilum Mre11-Rad50-Nbs1 complex bound to ATPyS type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Chaetomium thermophilum (fungus) Chaetomium thermophilum (fungus) |
-Macromolecule #1: Double-strand break repair protein
| Macromolecule | Name: Double-strand break repair protein / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Thermochaetoides thermophila (fungus) / Strain: DSM 1495 / CBS 144.50 / IMI 039719 Thermochaetoides thermophila (fungus) / Strain: DSM 1495 / CBS 144.50 / IMI 039719 |
| Molecular weight | Theoretical: 83.036273 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MPQTAGPDTI RILVSTDNHV GYEERDPIRK DDSWRTFDEI MQLARTKDVD MVLLGGDLFH DNKPSRKAMY QVMRSLRKNC LGMKPCELE FLSDPAEVFE GAFPHVNYYD PDINVSIPVF SIHGNHDDPS GDGHLCSLDL LQVAGLVNYF GRVPEADNIH V KPILLQKG ...String: MPQTAGPDTI RILVSTDNHV GYEERDPIRK DDSWRTFDEI MQLARTKDVD MVLLGGDLFH DNKPSRKAMY QVMRSLRKNC LGMKPCELE FLSDPAEVFE GAFPHVNYYD PDINVSIPVF SIHGNHDDPS GDGHLCSLDL LQVAGLVNYF GRVPEADNIH V KPILLQKG KTKLALYGMS NVRDERIHRT FRDNKVRFYR PSQQTGDWFN LLTLHQNHYA HTPTGYLSEN MLPDFLDLVI WG HEHECLI DPKKNPETGF HVMQPGSSIA TSLVPGEAVP KHIAILSITG KSFEVEKIPL RTVRPFVIRE ITLATDKRFK GLE KKQDNR QEVTKRLMQI VEEMIAEANE MWRSLHEDSQ DDEDEEQPLP LIRLKVEYSS PEGTKFEVEN PQRFSNRFAG KVAN QNDVV HFYRKKTGTT RKPKEGKREL PEGIAEALED SDSISVDALV QEFFAQQSLK ILPQAPFGDA VNQFVSKDDK HAVEM FVMD SLSSQVRGLL QLDDDKINEG LDSHIEDFRK VMEKNFLSGQ QKQAQRRRRF KEKPEGWDSD LNGHWTLQPE AIEELS SSP EPAKEGGRVR PASRITVGDE DNLFEEEEFV QKTTAKRAPT TRATRKTAAA TRATTATKAS APAKKSIAAP RGRKRAN PF QDSAEEEEDV IMDDDDDYKP APPVKAPPPK PARETQTRGA PKTRQTTLNF SQAERPTRTT QKAIEISDDE ISEDDAFE S MPARKSKRY UniProtKB: Double-strand break repair protein MRE11 |
-Macromolecule #2: DH domain-containing protein
| Macromolecule | Name: DH domain-containing protein / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Thermochaetoides thermophila (fungus) / Strain: DSM 1495 / CBS 144.50 / IMI 039719 Thermochaetoides thermophila (fungus) / Strain: DSM 1495 / CBS 144.50 / IMI 039719 |
| Molecular weight | Theoretical: 152.229906 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MSKIEKLSIL GVRSFGPHHP ETIAFNTPLT LIVGYNGSGK TTVIECLKYA TTGELPPNST RNGAFIHDPD LVGEKEVRAQ VKLSFRSTI GESYVVTRNI QLLVQRNNKR TQKTLEGSLL LRNNGERTVI STRVAELDKL VSEKLGVPPA ILDAVIFCHQ D DSLWPMSE ...String: MSKIEKLSIL GVRSFGPHHP ETIAFNTPLT LIVGYNGSGK TTVIECLKYA TTGELPPNST RNGAFIHDPD LVGEKEVRAQ VKLSFRSTI GESYVVTRNI QLLVQRNNKR TQKTLEGSLL LRNNGERTVI STRVAELDKL VSEKLGVPPA ILDAVIFCHQ D DSLWPMSE PAALKKRFDE IFEAQKYTKV IENIRLLKKK KGDELKILKE REVQDKANKE RAEKVDRLMA QLTREILEAR EK CNELSKQ MEEESAKIKD KYEQANSFLK IMNDLQTKTE KLEYKKDAIV ELRSRIEELP DPDEVLRNTL DEYEQTINRI VAD RDHKAA QFHDLQAELK SARDQHTAKA AEQGKHQSDK EKYERQLVAR ERMIREAAER HEIRGYNGDL DDRRIAIFNE RIQK ILNDK RRELERLQRE NQEELDRKTA VIAERESRKQ SVIRDRKAAK DRIISLGKDM ASIQGELSSI DIDEGTEEML RAEMK ELQA RIEAAKADEQ NANLDAQIKE VNEEIWKLES LSAKLARELV ECTRLASERA QLDLRRKQLA ERKRELEIMT NTWNEQ FST LLGEGWRPET LERDFSDVLK QQQLLVGEHR KKKDATQQEL KQAEYQLSNA RNLHNKLTNE MEACMRAVQT AMKEARD LD SAPPVDEYIT MLETDEKELA EVETALKLYD ELKKHYSTIK DRALRFNKCY ICDRDFTNQE AAKTRLLEKV AKRLGDEE K KELLEDQAAF MKSLDILRAV RVKYDTYQRL SSELPQLSRE IDSETNRRED LVRRLEDQDL AFREADNKLQ EMETLNKHV MKITQLLKDI SDAEKQVERS QQLSNIETRS ADEINEEQTT CAEQTRAAQA KLTKLTAEKQ RLKDLVRQLE VERLQLENKI SSAVQQLER KKRLQESIAR HKEDQNQARN AVQEADEELE RLEPEIAGAR AALDEARQAC RAKEQKVAAE RDAIAQTVSE L NMINSEIQ EYLDRGGPSS LAANQRAIAN LETQMANLEG EMRELTVQIN KLNKEIDNSD AKKRNIADNL TYRKNLREKD AL EREIAEL EARNAQEDYD RLIKEAHYLE AHRSKLNADR ERLMGMMSTK DEEFRRLNEE YELDLKDAKA KYKETHIKVE TTK AAIEDL GRGMAAVDHA IMQYHSKMME QINRTIAELW QSTYQGTDID TIQIRSDVES TTSSDSGTRR NYNYRVSMVK GDTE MDMRG RCSAGQKVLA SIIIRLALAE SFCANCGLIA LDEPTTNLDS DNIRSLAESL HGIIKARQAQ GNLQLIVITH DEEFL KYMQ CSDFCDDFYR VKRDEKQNSV IVRESITRIT E UniProtKB: DNA repair protein RAD50 |
-Macromolecule #3: FHA domain-containing protein
| Macromolecule | Name: FHA domain-containing protein / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Thermochaetoides thermophila (fungus) / Strain: DSM 1495 / CBS 144.50 / IMI 039719 Thermochaetoides thermophila (fungus) / Strain: DSM 1495 / CBS 144.50 / IMI 039719 |
| Molecular weight | Theoretical: 105.980812 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MWILESELFD GKRLWLRPGK TYLFGRTVAE AGQLTISDKT VSRKHLTIHI DNVPEGGGRN LRSRSNVIVE DLESKKGTLV NGVQIRGQK TTLTEDVNEI KLGLCPKTLK IRWHPIVLSF SFTSKELRAD PWTNLRDSLE QLDIKYSAEY EPTTTHVVSK K RNTSKGLQ ...String: MWILESELFD GKRLWLRPGK TYLFGRTVAE AGQLTISDKT VSRKHLTIHI DNVPEGGGRN LRSRSNVIVE DLESKKGTLV NGVQIRGQK TTLTEDVNEI KLGLCPKTLK IRWHPIVLSF SFTSKELRAD PWTNLRDSLE QLDIKYSAEY EPTTTHVVSK K RNTSKGLQ ALINGRYIVT DSFINAIVQA TEIPEGEEGA SSALEQDFEA NWPNPLDHLP PRGEEPGNHT TETYAPDARR QE VFDGYTF IFYEKKQYDN LFPAISAGKG KALLKEVVPN RTRVDEFVRY VKSVAGEKGL GSFEDGSEGK GVVVVRYTPK GED SAWYAE FFTKFAQQLD HRPIDQKEFL EAILACDASM LRRPLEAMSQ PVSVSASVEP QSSEKVRPAV EDRKEVEQSA PKQL QPSAE VPATEESAPA PHRRERRTGR SRFKGFDFDD DDIIIETPQA QSSTQVPALP QVPSASQDSL FVSQREPSLA PSEPM LEEE APCNTRTTRQ THRKRVLSPL PEHDNSALLD EIAPITAAVK RRRIEAGQDP VPPLPEPEPE REDEDVEMVE ESPPRK GKK GAATTAKGKG KKIKQEDEEN VLELARRRRE EAEAAAAAER QRLAQLGDDD IDYAAIRRLH IIEEIEVRQP EPHGPNR TR EQDIADGRWD PRWNGRKNFK RFRRQGETGV RMPVQSVIVP LEEVRTKEYG IGDDYWLEDE EGRVPRRPKE TQTQERST I GSVRDGSGFA AAAASGKGKE KDKENEKEVG RPGSSAAAAK QRSKPAPRRT VLTLDSSDED EDEPSPHAPG IDTISDSEP EVVSSFPSVI PASEPSRSRA AKAAERANAL RSSAHSSQSQ TQQHRESQLS TGSSKIQLTL APGSSSLSFS RSGTAAGRNE NGKRPFGSF VSGESTASGR GMSVESGSVR GESASKRQKQ GSSGGGSFLA TRRKDDGSEE ESEDDELKFR FGRRR UniProtKB: FHA domain-containing protein |
-Macromolecule #4: MANGANESE (II) ION
| Macromolecule | Name: MANGANESE (II) ION / type: ligand / ID: 4 / Number of copies: 4 / Formula: MN |
|---|---|
| Molecular weight | Theoretical: 54.938 Da |
-Macromolecule #5: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 5 / Number of copies: 2 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Macromolecule #6: PHOSPHOTHIOPHOSPHORIC ACID-ADENYLATE ESTER
| Macromolecule | Name: PHOSPHOTHIOPHOSPHORIC ACID-ADENYLATE ESTER / type: ligand / ID: 6 / Number of copies: 2 / Formula: AGS |
|---|---|
| Molecular weight | Theoretical: 523.247 Da |
| Chemical component information |  ChemComp-AGS: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.27 mg/mL |
|---|---|
| Buffer | pH: 7.6 |
| Grid | Model: Quantifoil R2/1 / Material: COPPER / Mesh: 200 / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 43.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.8000000000000003 µm / Nominal defocus min: 1.0 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 4.0 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 288443 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller