[English] 日本語
 Yorodumi
Yorodumi- EMDB-10451: ER microsome released with the code in (Martinez-Sanchez et al., ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-10451 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | ER microsome released with the code in (Martinez-Sanchez et al., Nature Methods) | |||||||||
 Map data Map data | ER microsome used in the code released in (Martinez-Sanchez et al., Nature Methods) | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  | |||||||||
| Method | electron tomography / cryo EM | |||||||||
 Authors Authors | Martinez-Sanchez A / Lucic V / Pfeffer S / Forster F | |||||||||
| Funding support |  Spain, 1 items Spain, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Methods / Year: 2020 Journal: Nat Methods / Year: 2020Title: Template-free detection and classification of membrane-bound complexes in cryo-electron tomograms. Authors: Antonio Martinez-Sanchez / Zdravko Kochovski / Ulrike Laugks / Johannes Meyer Zum Alten Borgloh / Saikat Chakraborty / Stefan Pfeffer / Wolfgang Baumeister / Vladan Lučić /  Abstract: With faithful sample preservation and direct imaging of fully hydrated biological material, cryo-electron tomography provides an accurate representation of molecular architecture of cells. However, ...With faithful sample preservation and direct imaging of fully hydrated biological material, cryo-electron tomography provides an accurate representation of molecular architecture of cells. However, detection and precise localization of macromolecular complexes within cellular environments is aggravated by the presence of many molecular species and molecular crowding. We developed a template-free image processing procedure for accurate tracing of complex networks of densities in cryo-electron tomograms, a comprehensive and automated detection of heterogeneous membrane-bound complexes and an unsupervised classification (PySeg). Applications to intact cells and isolated endoplasmic reticulum (ER) allowed us to detect and classify small protein complexes. This classification provided sufficiently homogeneous particle sets and initial references to allow subsequent de novo subtomogram averaging. Spatial distribution analysis showed that ER complexes have different localization patterns forming nanodomains. Therefore, this procedure allows a comprehensive detection and structural analysis of complexes in situ. #1:  Journal: Nature Communications / Year: 2015 Journal: Nature Communications / Year: 2015Title: Structure of the native Sec61 protein-conducting channel Authors: Pfeffer S / Forster F | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_10451.map.gz emd_10451.map.gz | 4.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-10451-v30.xml emd-10451-v30.xml emd-10451.xml emd-10451.xml | 12.9 KB 12.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_10451.png emd_10451.png | 185.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-10451 http://ftp.pdbj.org/pub/emdb/structures/EMD-10451 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10451 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10451 | HTTPS FTP |
-Validation report
| Summary document |  emd_10451_validation.pdf.gz emd_10451_validation.pdf.gz | 217.4 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_10451_full_validation.pdf.gz emd_10451_full_validation.pdf.gz | 216.5 KB | Display | |
| Data in XML |  emd_10451_validation.xml.gz emd_10451_validation.xml.gz | 4.3 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-10451 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-10451 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-10451 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-10451 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_10451.map.gz / Format: CCP4 / Size: 5.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_10451.map.gz / Format: CCP4 / Size: 5.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | ER microsome used in the code released in (Martinez-Sanchez et al., Nature Methods) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 10.48 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
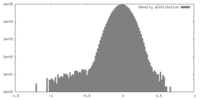
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Representative tomogram of a canine pancreatic rough Endoplasmic ...
| Entire | Name: Representative tomogram of a canine pancreatic rough Endoplasmic Reticulum vesicle. |
|---|---|
| Components |
|
-Supramolecule #1: Representative tomogram of a canine pancreatic rough Endoplasmic ...
| Supramolecule | Name: Representative tomogram of a canine pancreatic rough Endoplasmic Reticulum vesicle. type: organelle_or_cellular_component / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | electron tomography |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2.00 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.6 Component:
| ||||||||||||
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 70 % / Instrument: FEI VITROBOT MARK IV / Details: Blot 3 seconds before plunging.. | ||||||||||||
| Sectioning | Other: NO SECTIONING | ||||||||||||
| Fiducial marker | Manufacturer: Aurion, Wageningen, The Netherlands / Diameter: 10 nm |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 1.5 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: OTHER / Imaging mode: OTHER / Cs: 2.7 mm / Nominal defocus max: 4.0 µm / Nominal defocus min: 3.0 µm / Nominal magnification: 42000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | Reconstruction of a single microsome after two binning steps (from 2.62A/pixel to 10.48A/pixel) |
|---|---|
| Final reconstruction | Software - Name: PyTom / Number images used: 61 |
| CTF correction | Software - Name: PyTom |
 Movie
Movie Controller
Controller













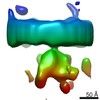
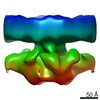




 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)