+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7uai | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
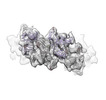
| Title | Meprin alpha helix in complex with fetuin-B | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords |  ONCOPROTEIN / ONCOPROTEIN /  Metalloprotease / Metalloprotease /  complex / helical / complex / helical /  extracellular extracellular | |||||||||
| Function / homology |  Function and homology information Function and homology information meprin A / meprin A /  meprin A complex / epidermal growth factor receptor ligand maturation / metalloendopeptidase inhibitor activity / metallodipeptidase activity / negative regulation of endopeptidase activity / binding of sperm to zona pellucida / cysteine-type endopeptidase inhibitor activity / meprin A complex / epidermal growth factor receptor ligand maturation / metalloendopeptidase inhibitor activity / metallodipeptidase activity / negative regulation of endopeptidase activity / binding of sperm to zona pellucida / cysteine-type endopeptidase inhibitor activity /  endopeptidase inhibitor activity / signaling receptor ligand precursor processing ... endopeptidase inhibitor activity / signaling receptor ligand precursor processing ... meprin A / meprin A /  meprin A complex / epidermal growth factor receptor ligand maturation / metalloendopeptidase inhibitor activity / metallodipeptidase activity / negative regulation of endopeptidase activity / binding of sperm to zona pellucida / cysteine-type endopeptidase inhibitor activity / meprin A complex / epidermal growth factor receptor ligand maturation / metalloendopeptidase inhibitor activity / metallodipeptidase activity / negative regulation of endopeptidase activity / binding of sperm to zona pellucida / cysteine-type endopeptidase inhibitor activity /  endopeptidase inhibitor activity / signaling receptor ligand precursor processing / single fertilization / endopeptidase inhibitor activity / signaling receptor ligand precursor processing / single fertilization /  metalloendopeptidase activity / metalloendopeptidase activity /  metallopeptidase activity / metallopeptidase activity /  extracellular space / extracellular exosome / zinc ion binding / extracellular region / extracellular space / extracellular exosome / zinc ion binding / extracellular region /  plasma membrane plasma membraneSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.8 Å cryo EM / Resolution: 2.8 Å | |||||||||
 Authors Authors | Bayly-Jones, C. / Lupton, C.J. / Fritz, C. / Schlenzig, D. / Whisstock, J.C. | |||||||||
| Funding support |  Germany, Germany,  Australia, 2items Australia, 2items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Helical ultrastructure of the metalloprotease meprin α in complex with a small molecule inhibitor. Authors: Charles Bayly-Jones / Christopher J Lupton / Claudia Fritz / Hariprasad Venugopal / Daniel Ramsbeck / Michael Wermann / Christian Jäger / Alex de Marco / Stephan Schilling / Dagmar ...Authors: Charles Bayly-Jones / Christopher J Lupton / Claudia Fritz / Hariprasad Venugopal / Daniel Ramsbeck / Michael Wermann / Christian Jäger / Alex de Marco / Stephan Schilling / Dagmar Schlenzig / James C Whisstock /   Abstract: The zinc-dependent metalloprotease meprin α is predominantly expressed in the brush border membrane of proximal tubules in the kidney and enterocytes in the small intestine and colon. In normal ...The zinc-dependent metalloprotease meprin α is predominantly expressed in the brush border membrane of proximal tubules in the kidney and enterocytes in the small intestine and colon. In normal tissue homeostasis meprin α performs key roles in inflammation, immunity, and extracellular matrix remodelling. Dysregulated meprin α is associated with acute kidney injury, sepsis, urinary tract infection, metastatic colorectal carcinoma, and inflammatory bowel disease. Accordingly, meprin α is the target of drug discovery programs. In contrast to meprin β, meprin α is secreted into the extracellular space, whereupon it oligomerises to form giant assemblies and is the largest extracellular protease identified to date (~6 MDa). Here, using cryo-electron microscopy, we determine the high-resolution structure of the zymogen and mature form of meprin α, as well as the structure of the active form in complex with a prototype small molecule inhibitor and human fetuin-B. Our data reveal that meprin α forms a giant, flexible, left-handed helical assembly of roughly 22 nm in diameter. We find that oligomerisation improves proteolytic and thermal stability but does not impact substrate specificity or enzymatic activity. Furthermore, structural comparison with meprin β reveal unique features of the active site of meprin α, and helical assembly more broadly. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7uai.cif.gz 7uai.cif.gz | 492.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7uai.ent.gz pdb7uai.ent.gz | 404.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7uai.json.gz 7uai.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ua/7uai https://data.pdbj.org/pub/pdb/validation_reports/ua/7uai ftp://data.pdbj.org/pub/pdb/validation_reports/ua/7uai ftp://data.pdbj.org/pub/pdb/validation_reports/ua/7uai | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  26426MC  7uabC  7uacC  7uaeC  7uafC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 2 types, 6 molecules EBCDAH
| #1: Protein |  / Endopeptidase-2 / N-benzoyl-L-tyrosyl-P-amino-benzoic acid hydrolase subunit alpha / PABA peptide ...Endopeptidase-2 / N-benzoyl-L-tyrosyl-P-amino-benzoic acid hydrolase subunit alpha / PABA peptide hydrolase / PPH alpha / Endopeptidase-2 / N-benzoyl-L-tyrosyl-P-amino-benzoic acid hydrolase subunit alpha / PABA peptide ...Endopeptidase-2 / N-benzoyl-L-tyrosyl-P-amino-benzoic acid hydrolase subunit alpha / PABA peptide hydrolase / PPH alphaMass: 67285.820 Da / Num. of mol.: 4 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: MEP1A / Plasmid: pMT/BiP/V5 / Cell line (production host): Schneider-2 / Production host: Homo sapiens (human) / Gene: MEP1A / Plasmid: pMT/BiP/V5 / Cell line (production host): Schneider-2 / Production host:   Drosophila melanogaster (fruit fly) / References: UniProt: Q16819, Drosophila melanogaster (fruit fly) / References: UniProt: Q16819,  meprin A meprin A#2: Protein | Mass: 42926.691 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: FETUB / Cell line (production host): HEK293 / Production host: Homo sapiens (human) / Gene: FETUB / Cell line (production host): HEK293 / Production host:   Homo sapiens (human) / References: UniProt: Q9UGM5 Homo sapiens (human) / References: UniProt: Q9UGM5 |
|---|
-Sugars , 4 types, 19 molecules 
| #3: Polysaccharide |  / Mass: 627.594 Da / Num. of mol.: 3 / Mass: 627.594 Da / Num. of mol.: 3Source method: isolated from a genetically manipulated source #4: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose  / Mass: 424.401 Da / Num. of mol.: 8 / Mass: 424.401 Da / Num. of mol.: 8Source method: isolated from a genetically manipulated source #5: Polysaccharide | beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1- ...beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-beta-D-glucopyranose  / Mass: 732.682 Da / Num. of mol.: 4 / Mass: 732.682 Da / Num. of mol.: 4Source method: isolated from a genetically manipulated source #8: Sugar | ChemComp-NAG /  N-Acetylglucosamine N-Acetylglucosamine |
|---|
-Non-polymers , 2 types, 12 molecules 


| #6: Chemical | ChemComp-CA / #7: Chemical | ChemComp-ZN / |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: FILAMENT / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Tetrameric portion of meprin alpha helix in complex with fetuin-B [subparticle localised reconstruction] Type: COMPLEX Details: Subparticle localised reconstruction of tetrameric region of recombinant, secreted helical meprin alpha in complex with fetuin-B Entity ID: #1-#2 / Source: RECOMBINANT | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 85 kDa/nm / Experimental value: NO | |||||||||||||||
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) | |||||||||||||||
| Source (recombinant) | Organism:   Drosophila (fruit flies) / Cell: Schneider-2 / Plasmid Drosophila (fruit flies) / Cell: Schneider-2 / Plasmid : pMT/BiP/V5 : pMT/BiP/V5 | |||||||||||||||
| Buffer solution | pH: 7.4 | |||||||||||||||
| Buffer component |
| |||||||||||||||
| Specimen | Conc.: 1 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES | |||||||||||||||
| Specimen support | Details: Pelco EasyGlow / Grid material: COPPER / Grid mesh size: 200 divisions/in. / Grid type: Quantifoil R2/2 | |||||||||||||||
Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 277.15 K / Details: 3 s blot, -3 force |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal magnification: 130000 X / Nominal defocus max: 2000 nm / Nominal defocus min: 500 nm / Cs Bright-field microscopy / Nominal magnification: 130000 X / Nominal defocus max: 2000 nm / Nominal defocus min: 500 nm / Cs : 2.7 mm : 2.7 mm |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Electron dose: 44.5 e/Å2 / Detector mode: COUNTING / Film or detector model: GATAN K3 (6k x 4k) / Num. of grids imaged: 1 / Num. of real images: 4068 |
| EM imaging optics | Energyfilter name : GIF Bioquantum : GIF Bioquantum |
| Image scans | Movie frames/image: 25 / Used frames/image: 1-25 |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.17.1_3660: / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Image processing | Details: Compressed to LZW TIFF. Motion corrected by MotionCor. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 519760 Details: Localised subparticles extrated after expansion of pseudo-helical symmetry | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry : C1 (asymmetric) : C1 (asymmetric) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
3D reconstruction | Resolution: 2.8 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 115951 / Algorithm: FOURIER SPACE / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT / Space: REAL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller









 PDBj
PDBj










