+Search query
-Structure paper
| Title | Structural basis for α-tubulin-specific and modification state-dependent glutamylation. |
|---|---|
| Journal, issue, pages | Nat Chem Biol, Year 2024 |
| Publish date | Apr 24, 2024 |
 Authors Authors | Kishore K Mahalingan / Danielle A Grotjahn / Yan Li / Gabriel C Lander / Elena A Zehr / Antonina Roll-Mecak /  |
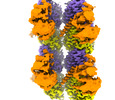
| PubMed Abstract | Microtubules have spatiotemporally complex posttranslational modification patterns. Tubulin tyrosine ligase-like (TTLL) enzymes introduce the most prevalent modifications on α-tubulin and β-tubulin. ...Microtubules have spatiotemporally complex posttranslational modification patterns. Tubulin tyrosine ligase-like (TTLL) enzymes introduce the most prevalent modifications on α-tubulin and β-tubulin. How TTLLs specialize for specific substrate recognition and ultimately modification-pattern generation is largely unknown. TTLL6, a glutamylase implicated in ciliopathies, preferentially modifies tubulin α-tails in microtubules. Cryo-electron microscopy, kinetic analysis and single-molecule biochemistry reveal an unprecedented quadrivalent recognition that ensures simultaneous readout of microtubule geometry and posttranslational modification status. By binding to a β-tubulin subunit, TTLL6 modifies the α-tail of the longitudinally adjacent tubulin dimer. Spanning two tubulin dimers along and across protofilaments (PFs) ensures fidelity of recognition of both the α-tail and the microtubule. Moreover, TTLL6 reads out and is stimulated by glutamylation of the β-tail of the laterally adjacent tubulin dimer, mediating crosstalk between α-tail and β-tail. This positive feedback loop can generate localized microtubule glutamylation patterns. Our work uncovers general principles that generate tubulin chemical and topographic complexity. |
 External links External links |  Nat Chem Biol / Nat Chem Biol /  PubMed:38658656 PubMed:38658656 |
| Methods | EM (single particle) |
| Resolution | 3.6 - 7.2 Å |
| Structure data | EMDB-41018: Microtubule-TTLL6 map  EMDB-41022: Map of Tubulin Tyrosine Ligase Like 6 EMDB-41090: Composite map of TTLL6 bound to unmodified human microtubule  EMDB-42884: Microtubule-TTLL6 map |
| Chemicals |  ChemComp-GTP:  ChemComp-MG:  ChemComp-G2P:  ChemComp-ATP: |
| Source |
|
 Keywords Keywords |  LIGASE / tubulin post-translational modifications / LIGASE / tubulin post-translational modifications /  microtubules / TTLL / microtubules / TTLL /  tubulin / post-translational modifications / Tubulin Tyrosine Ligase Like family enzymes tubulin / post-translational modifications / Tubulin Tyrosine Ligase Like family enzymes |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers