+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-6555 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
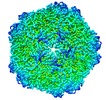
| タイトル | 2.9 Angstrom Resolution Cryo-EM 3-D Reconstruction of Close-packed PCV2 Virus-like Particles | |||||||||
 マップデータ マップデータ | Reconstruction of porcine circovirus 2 (PCV2) virus-like particles | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | de novo initial model / consensus criterion / gold-standard FSC / true FSC / cross-validation | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報viral capsid assembly / T=1 icosahedral viral capsid / endocytosis involved in viral entry into host cell / viral penetration into host nucleus / host cell nucleus / virion attachment to host cell / nucleus 類似検索 - 分子機能 | |||||||||
| 生物種 |   Porcine circovirus 2 (ウイルス) Porcine circovirus 2 (ウイルス) | |||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 2.9 Å | |||||||||
 データ登録者 データ登録者 | Liu Z / Guo F / Wang F / Li TC / Jiang W | |||||||||
 引用 引用 |  ジャーナル: Structure / 年: 2016 ジャーナル: Structure / 年: 2016タイトル: 2.9 Å Resolution Cryo-EM 3D Reconstruction of Close-Packed Virus Particles. 著者: Zheng Liu / Fei Guo / Feng Wang / Tian-Cheng Li / Wen Jiang /   要旨: Single-particle cryoelectron microscopy typically discards close-packed particle images as unusable data. Here, we report an image processing strategy and case study of obtaining near-atomic ...Single-particle cryoelectron microscopy typically discards close-packed particle images as unusable data. Here, we report an image processing strategy and case study of obtaining near-atomic resolution 3D reconstructions from close-packed particles. Multiple independent de novo initial models were constructed to determine and cross-validate the particle parameters. The particles with consistent views were further refined including not only Euler angles and center positions but also defocus, astigmatism, beam tilt, and overall and anisotropic magnification. We demonstrated this strategy with a 2.9 Å resolution reconstruction of a 1.67 MDa virus-like particle of a circovirus, PCV2, recorded on 86 photographic films. The map resolution was further validated with a phase-randomization test and local resolution assessment, and the atomic model was validated with MolProbity and EMRinger. Close-packed virus particles were thus shown not only to be useful for high-resolution 3D reconstructions but also to allow data collection at significantly improved throughput for near-atomic resolution reconstructions. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_6555.map.gz emd_6555.map.gz | 108 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-6555-v30.xml emd-6555-v30.xml emd-6555.xml emd-6555.xml | 12.5 KB 12.5 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_6555.png emd_6555.png | 546.1 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-6555 http://ftp.pdbj.org/pub/emdb/structures/EMD-6555 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6555 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6555 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_6555_validation.pdf.gz emd_6555_validation.pdf.gz | 367.6 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_6555_full_validation.pdf.gz emd_6555_full_validation.pdf.gz | 367.2 KB | 表示 | |
| XML形式データ |  emd_6555_validation.xml.gz emd_6555_validation.xml.gz | 7 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6555 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6555 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6555 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6555 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_6555.map.gz / 形式: CCP4 / 大きさ: 122.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_6555.map.gz / 形式: CCP4 / 大きさ: 122.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Reconstruction of porcine circovirus 2 (PCV2) virus-like particles | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : Porcine circovirus PCV2 virus-like particles
| 全体 | 名称: Porcine circovirus PCV2 virus-like particles |
|---|---|
| 要素 |
|
-超分子 #1000: Porcine circovirus PCV2 virus-like particles
| 超分子 | 名称: Porcine circovirus PCV2 virus-like particles / タイプ: sample / ID: 1000 / Number unique components: 1 |
|---|---|
| 分子量 | 実験値: 1.67 MDa / 理論値: 1.67 MDa |
-超分子 #1: Porcine circovirus 2
| 超分子 | 名称: Porcine circovirus 2 / タイプ: virus / ID: 1 / Name.synonym: PCV2 / NCBI-ID: 85708 / 生物種: Porcine circovirus 2 / Sci species strain: Yamagata / ウイルスタイプ: VIRUS-LIKE PARTICLE / ウイルス・単離状態: STRAIN / ウイルス・エンベロープ: No / ウイルス・中空状態: Yes / Syn species name: PCV2 |
|---|---|
| 宿主 | 生物種:  |
| Host system | 生物種:  Trichoplusia ni (イラクサキンウワバ) / 組換細胞: Tn5 / 組換プラスミド: AcPCV2-ORF2 Trichoplusia ni (イラクサキンウワバ) / 組換細胞: Tn5 / 組換プラスミド: AcPCV2-ORF2 |
| 分子量 | 実験値: 1.67 MDa / 理論値: 1.67 MDa |
| ウイルス殻 | Shell ID: 1 / 直径: 190 Å / T番号(三角分割数): 1 |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 3 mg/mL |
|---|---|
| 緩衝液 | pH: 7.4 / 詳細: 1x PBS |
| グリッド | 詳細: 400-mesh holey carbon grids (1.2/1.3 C-flat, Protochips) |
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 90 % / チャンバー内温度: 85 K / 装置: GATAN CRYOPLUNGE 3 / 手法: Blot for 5 seconds before plunging. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 温度 | 最低: 80 K / 最高: 100 K / 平均: 90 K |
| アライメント法 | Legacy - 非点収差: Objective lens astigmatism was corrected at 250,000 magnification using a quadrupole stigmator. |
| 日付 | 2011年2月22日 |
| 撮影 | カテゴリ: FILM / フィルム・検出器のモデル: KODAK SO-163 FILM デジタル化 - スキャナー: NIKON SUPER COOLSCAN 9000 デジタル化 - サンプリング間隔: 6.35 µm / 実像数: 141 / 平均電子線量: 25 e/Å2 / Od range: 1 / ビット/ピクセル: 16 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm / 最大 デフォーカス(公称値): 2.5 µm / 最小 デフォーカス(公称値): 0.2 µm / 倍率(公称値): 59000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 詳細 | The particles were selected using the e2boxer.py program in EMAN2. CTF parameters were determined using fitctf2.py in JSPR. |
|---|---|
| CTF補正 | 詳細: Each particle |
| 最終 再構成 | アルゴリズム: OTHER / 解像度のタイプ: BY AUTHOR / 解像度: 2.9 Å / 解像度の算出法: OTHER / ソフトウェア - 名称: JSPR, EMAN2 詳細: For 3D reconstruction, whole datasets were divided into even and odd halves and the initial de novo models and subsequent iterative refinements were all independently performed for each half ...詳細: For 3D reconstruction, whole datasets were divided into even and odd halves and the initial de novo models and subsequent iterative refinements were all independently performed for each half dataset. Particles were selected from scanned micrograph images using e2boxer.py in EMAN2. The TEM instrument contrast transfer function parameters were determined automatically using fitctf2.py in JSPR and were then visually validated using the EMAN ctfit program. The datasets were then divided into two subsets (even and odd) and processed completely independently, including both de novo initial models and refinements. The images were first binned 4x to obtain initial models and particle parameters assuming icosahedral symmetry. De novo initial models were built using the random model approach. Random subsets of particles were assigned random initial orientations and iteratively refined until convergence. Multi-model competitive refinements were used to choose the winning model (with most assigned particles) as corrective initial models for subsequent refinement. Particles with inconsistent/unstable view parameters in the initial refinements were excluded in further image processing. The orientation and center parameters were then transferred to the un-binned images for high-resolution refinements which included Simplex method-based orientation/center optimization and grid search-based refinement of defocus, astigmatism, beam tilt, and overall and anisotropic magnification of the images. All image refinement and reconstructions were performed with JSPR software that was built on EMAN2 and EMAN library functions and programs. 使用した粒子像数: 50352 |
 ムービー
ムービー コントローラー
コントローラー