+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-0259 | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Paired helical filament from sporadic Alzheimer's disease brain タウオパチー タウオパチー | ||||||||||||||||||
 マップデータ マップデータ | |||||||||||||||||||
 試料 試料 |
| ||||||||||||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報plus-end-directed organelle transport along microtubule /  軸索輸送 / histone-dependent DNA binding / 軸索輸送 / histone-dependent DNA binding /  neurofibrillary tangle assembly / positive regulation of diacylglycerol kinase activity / negative regulation of establishment of protein localization to mitochondrion / neurofibrillary tangle assembly / positive regulation of diacylglycerol kinase activity / negative regulation of establishment of protein localization to mitochondrion /  neurofibrillary tangle / positive regulation of protein localization to synapse / microtubule lateral binding / neurofibrillary tangle / positive regulation of protein localization to synapse / microtubule lateral binding /  tubulin complex ...plus-end-directed organelle transport along microtubule / tubulin complex ...plus-end-directed organelle transport along microtubule /  軸索輸送 / histone-dependent DNA binding / 軸索輸送 / histone-dependent DNA binding /  neurofibrillary tangle assembly / positive regulation of diacylglycerol kinase activity / negative regulation of establishment of protein localization to mitochondrion / neurofibrillary tangle assembly / positive regulation of diacylglycerol kinase activity / negative regulation of establishment of protein localization to mitochondrion /  neurofibrillary tangle / positive regulation of protein localization to synapse / microtubule lateral binding / neurofibrillary tangle / positive regulation of protein localization to synapse / microtubule lateral binding /  tubulin complex / tubulin complex /  phosphatidylinositol bisphosphate binding / main axon / regulation of long-term synaptic depression / negative regulation of kinase activity / negative regulation of tubulin deacetylation / generation of neurons / regulation of chromosome organization / positive regulation of protein localization / rRNA metabolic process / internal protein amino acid acetylation / phosphatidylinositol bisphosphate binding / main axon / regulation of long-term synaptic depression / negative regulation of kinase activity / negative regulation of tubulin deacetylation / generation of neurons / regulation of chromosome organization / positive regulation of protein localization / rRNA metabolic process / internal protein amino acid acetylation /  regulation of mitochondrial fission / regulation of mitochondrial fission /  lipoprotein particle binding / intracellular distribution of mitochondria / axonal transport of mitochondrion / axon development / lipoprotein particle binding / intracellular distribution of mitochondria / axonal transport of mitochondrion / axon development /  central nervous system neuron development / central nervous system neuron development /  regulation of microtubule polymerization / regulation of microtubule polymerization /  微小管 / minor groove of adenine-thymine-rich DNA binding / negative regulation of mitochondrial membrane potential / 微小管 / minor groove of adenine-thymine-rich DNA binding / negative regulation of mitochondrial membrane potential /  dynactin binding / glial cell projection / dynactin binding / glial cell projection /  apolipoprotein binding / protein polymerization / negative regulation of mitochondrial fission / apolipoprotein binding / protein polymerization / negative regulation of mitochondrial fission /  axolemma / Caspase-mediated cleavage of cytoskeletal proteins / regulation of microtubule polymerization or depolymerization / positive regulation of axon extension / supramolecular fiber organization / Activation of AMPK downstream of NMDARs / regulation of microtubule cytoskeleton organization / axolemma / Caspase-mediated cleavage of cytoskeletal proteins / regulation of microtubule polymerization or depolymerization / positive regulation of axon extension / supramolecular fiber organization / Activation of AMPK downstream of NMDARs / regulation of microtubule cytoskeleton organization /  stress granule assembly / cytoplasmic microtubule organization / regulation of cellular response to heat / regulation of calcium-mediated signaling / axon cytoplasm / positive regulation of microtubule polymerization / cellular response to brain-derived neurotrophic factor stimulus / somatodendritic compartment / stress granule assembly / cytoplasmic microtubule organization / regulation of cellular response to heat / regulation of calcium-mediated signaling / axon cytoplasm / positive regulation of microtubule polymerization / cellular response to brain-derived neurotrophic factor stimulus / somatodendritic compartment /  synapse assembly / synapse assembly /  phosphatidylinositol binding / nuclear periphery / cellular response to nerve growth factor stimulus / positive regulation of superoxide anion generation / protein phosphatase 2A binding / phosphatidylinositol binding / nuclear periphery / cellular response to nerve growth factor stimulus / positive regulation of superoxide anion generation / protein phosphatase 2A binding /  regulation of autophagy / astrocyte activation / synapse organization / microglial cell activation / response to lead ion / regulation of autophagy / astrocyte activation / synapse organization / microglial cell activation / response to lead ion /  Hsp90 protein binding / Hsp90 protein binding /  regulation of synaptic plasticity / PKR-mediated signaling / protein homooligomerization / cytoplasmic ribonucleoprotein granule / regulation of synaptic plasticity / PKR-mediated signaling / protein homooligomerization / cytoplasmic ribonucleoprotein granule /  記憶 / microtubule cytoskeleton organization / cellular response to reactive oxygen species / 記憶 / microtubule cytoskeleton organization / cellular response to reactive oxygen species /  SH3 domain binding / neuron projection development / activation of cysteine-type endopeptidase activity involved in apoptotic process / microtubule cytoskeleton / protein-macromolecule adaptor activity / SH3 domain binding / neuron projection development / activation of cysteine-type endopeptidase activity involved in apoptotic process / microtubule cytoskeleton / protein-macromolecule adaptor activity /  single-stranded DNA binding / cell-cell signaling / cellular response to heat / single-stranded DNA binding / cell-cell signaling / cellular response to heat /  cell body / cell body /  actin binding / actin binding /  成長円錐 / protein-folding chaperone binding / 成長円錐 / protein-folding chaperone binding /  double-stranded DNA binding / double-stranded DNA binding /  microtubule binding / microtubule binding /  微小管 / amyloid fibril formation / sequence-specific DNA binding / 微小管 / amyloid fibril formation / sequence-specific DNA binding /  樹状突起スパイン / learning or memory / neuron projection / nuclear speck / 樹状突起スパイン / learning or memory / neuron projection / nuclear speck /  脂質ラフト / 脂質ラフト /  神経繊維 / negative regulation of gene expression / 神経繊維 / negative regulation of gene expression /  樹状突起 / neuronal cell body / DNA damage response / 樹状突起 / neuronal cell body / DNA damage response /  protein kinase binding / protein kinase binding /  enzyme binding / enzyme binding /  ミトコンドリア / ミトコンドリア /  DNA binding DNA binding類似検索 - 分子機能 | ||||||||||||||||||
| 生物種 |   Homo sapiens (ヒト) / Homo sapiens (ヒト) /   Human (ヒト) Human (ヒト) | ||||||||||||||||||
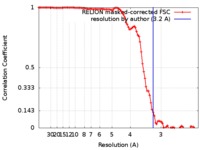
| 手法 | らせん対称体再構成法 /  クライオ電子顕微鏡法 / 解像度: 3.2 Å クライオ電子顕微鏡法 / 解像度: 3.2 Å | ||||||||||||||||||
 データ登録者 データ登録者 | Falcon B / Zhang W / Schweighauser M / Murzin AG / Vidal R / Garringer HJ / Ghetti B / Scheres SHW / Goedert M | ||||||||||||||||||
| 資金援助 |  英国, 英国,  米国, 5件 米国, 5件
| ||||||||||||||||||
 引用 引用 |  ジャーナル: Acta Neuropathol / 年: 2018 ジャーナル: Acta Neuropathol / 年: 2018タイトル: Tau filaments from multiple cases of sporadic and inherited Alzheimer's disease adopt a common fold. 著者: Benjamin Falcon / Wenjuan Zhang / Manuel Schweighauser / Alexey G Murzin / Ruben Vidal / Holly J Garringer / Bernardino Ghetti / Sjors H W Scheres / Michel Goedert /   要旨: The ordered assembly of tau protein into abnormal filaments is a defining characteristic of Alzheimer's disease (AD) and other neurodegenerative disorders. It is not known if the structures of tau ...The ordered assembly of tau protein into abnormal filaments is a defining characteristic of Alzheimer's disease (AD) and other neurodegenerative disorders. It is not known if the structures of tau filaments vary within, or between, the brains of individuals with AD. We used a combination of electron cryo-microscopy (cryo-EM) and immuno-gold negative-stain electron microscopy (immuno-EM) to determine the structures of paired helical filaments (PHFs) and straight filaments (SFs) from the frontal cortex of 17 cases of AD (15 sporadic and 2 inherited) and 2 cases of atypical AD (posterior cortical atrophy). The high-resolution structures of PHFs and SFs from the frontal cortex of 3 cases of AD, 2 sporadic and 1 inherited, were determined by cryo-EM. We also used immuno-EM to study the PHFs and SFs from a number of cortical and subcortical brain regions. PHFs outnumbered SFs in all AD cases. By cryo-EM, PHFs and SFs were made of two C-shaped protofilaments with a combined cross-β/β-helix structure, as described previously for one case of AD. The higher resolution structures obtained here showed two additional amino acids at each end of the protofilament. The immuno-EM findings, which indicated the presence of repeats 3 and 4, but not of the N-terminal regions of repeats 1 and 2, of tau in the filament cores of all AD cases, were consistent with the cryo-EM results. These findings show that there is no significant variation in tau filament structures between individuals with AD. This knowledge will be crucial for understanding the mechanisms that underlie tau filament formation and for developing novel diagnostics and therapies. | ||||||||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_0259.map.gz emd_0259.map.gz | 27.4 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-0259-v30.xml emd-0259-v30.xml emd-0259.xml emd-0259.xml | 17.8 KB 17.8 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_0259_fsc.xml emd_0259_fsc.xml | 9.6 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_0259.png emd_0259.png | 70.2 KB | ||
| マスクデータ |  emd_0259_msk_1.map emd_0259_msk_1.map | 75.1 MB |  マスクマップ マスクマップ | |
| その他 |  emd_0259_half_map_1.map.gz emd_0259_half_map_1.map.gz emd_0259_half_map_2.map.gz emd_0259_half_map_2.map.gz | 58.7 MB 58.7 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-0259 http://ftp.pdbj.org/pub/emdb/structures/EMD-0259 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0259 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0259 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  6hreMC  0260C  6hrfC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | |
| 電子顕微鏡画像生データ |  EMPIAR-10230 (タイトル: Cryo electron microscopy of tau filaments from sporadic Alzheimer's disease brain EMPIAR-10230 (タイトル: Cryo electron microscopy of tau filaments from sporadic Alzheimer's disease brainData size: 27.0 Data #1: Dose-weighted micrographs of tau filaments from sporadic Alzheimer's disease brain (case 2) [micrographs - single frame]) |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_0259.map.gz / 形式: CCP4 / 大きさ: 75.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_0259.map.gz / 形式: CCP4 / 大きさ: 75.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ボクセルのサイズ |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
-マスク #1
| ファイル |  emd_0259_msk_1.map emd_0259_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: Half map 1
| ファイル | emd_0259_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half map 1 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: Half map 2
| ファイル | emd_0259_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half map 2 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Tau filaments extracted from the frontotemporal cortex of a patie...
| 全体 | 名称: Tau filaments extracted from the frontotemporal cortex of a patient with sporadic Alzheimer's disease |
|---|---|
| 要素 |
|
-超分子 #1: Tau filaments extracted from the frontotemporal cortex of a patie...
| 超分子 | 名称: Tau filaments extracted from the frontotemporal cortex of a patient with sporadic Alzheimer's disease タイプ: tissue / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:   Homo sapiens (ヒト) / 器官: Brain / 組織: Frontotemporal cortex Homo sapiens (ヒト) / 器官: Brain / 組織: Frontotemporal cortex |
-分子 #1: Microtubule-associated protein tau
| 分子 | 名称: Microtubule-associated protein tau / タイプ: protein_or_peptide / ID: 1 / コピー数: 6 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:   Human (ヒト) / 組織: Brain Human (ヒト) / 組織: Brain |
| 分子量 | 理論値: 45.919871 KDa |
| 配列 | 文字列: MAEPRQEFEV MEDHAGTYGL GDRKDQGGYT MHQDQEGDTD AGLKESPLQT PTEDGSEEPG SETSDAKSTP TAEDVTAPLV DEGAPGKQA AAQPHTEIPE GTTAEEAGIG DTPSLEDEAA GHVTQARMVS KSKDGTGSDD KKAKGADGKT KIATPRGAAP P GQKGQANA ...文字列: MAEPRQEFEV MEDHAGTYGL GDRKDQGGYT MHQDQEGDTD AGLKESPLQT PTEDGSEEPG SETSDAKSTP TAEDVTAPLV DEGAPGKQA AAQPHTEIPE GTTAEEAGIG DTPSLEDEAA GHVTQARMVS KSKDGTGSDD KKAKGADGKT KIATPRGAAP P GQKGQANA TRIPAKTPPA PKTPPSSGEP PKSGDRSGYS SPGSPGTPGS RSRTPSLPTP PTREPKKVAV VRTPPKSPSS AK SRLQTAP VPMPDLKNVK SKIGSTENLK HQPGGGKVQI INKKLDLSNV QSKCGSKDNI KHVPGGGSVQ IVYKPVDLSK VTS KCGSLG NIHHKPGGGQ VEVKSEKLDF KDRVQSKIGS LDNITHVPGG GNKKIETHKL TFRENAKAKT DHGAEIVYKS PVVS GDTSP RHLSNVSSTG SIDMVDSPQL ATLADEVSAS LAKQGL |
-実験情報
-構造解析
| 手法 |  クライオ電子顕微鏡法 クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | らせん対称体再構成法 |
| 試料の集合状態 | tissue |
- 試料調製
試料調製
| 濃度 | 0.5 mg/mL | ||||||
|---|---|---|---|---|---|---|---|
| 緩衝液 | pH: 7.4 構成要素:
| ||||||
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 277 K / 装置: FEI VITROBOT MARK IV | ||||||
| 詳細 | Dispersed filaments |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm Bright-field microscopy / Cs: 2.7 mm最大 デフォーカス(公称値): 2.8000000000000003 µm 最小 デフォーカス(公称値): 1.7 µm |
| 特殊光学系 | エネルギーフィルター - 名称: GIF Quantum LS |
| 撮影 | フィルム・検出器のモデル: GATAN K2 QUANTUM (4k x 4k) 検出モード: COUNTING / 平均電子線量: 1.2 e/Å2 |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
-原子モデル構築 1
| 詳細 | Fourier-space refinement of the complete atomic model against the narrow Pick filament map was performed in REFMAC. A stack of three consecutive monomers was refined to preserve nearest-neighbour interactions for the middle chain. |
|---|---|
| 精密化 | 空間: RECIPROCAL / プロトコル: AB INITIO MODEL / 温度因子: 72.8 / 当てはまり具合の基準: Fourier shell correlation |
| 得られたモデル |  PDB-6hre: |
 ムービー
ムービー コントローラー
コントローラー
















 Z
Z Y
Y X
X