+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 9ojn | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of Mycobacterium smegmatis EtfD | |||||||||||||||||||||||||||
 Components Components | Iron-sulfur-binding reductase | |||||||||||||||||||||||||||
 Keywords Keywords | OXIDOREDUCTASE / membrane protein / electron transport chain / beta oxidation | |||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology information4 iron, 4 sulfur cluster binding / oxidoreductase activity / metal ion binding / plasma membrane Similarity search - Function | |||||||||||||||||||||||||||
| Biological species |  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria) | |||||||||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.2 Å | |||||||||||||||||||||||||||
 Authors Authors | Courbon, G.M. / Rubinstein, J.L. | |||||||||||||||||||||||||||
| Funding support |  Canada, 1items Canada, 1items
| |||||||||||||||||||||||||||
 Citation Citation |  Journal: EMBO J / Year: 2026 Journal: EMBO J / Year: 2026Title: Structural basis for EtfD-mediated coupling of β-oxidation and the respiratory chain in mycobacteria. Authors: Gautier M Courbon / Vadim Makarov / Stewart T Cole / Dirk Schnapinger / Sabine Ehrt / John L Rubinstein /     Abstract: Targeting β-oxidation has been proposed as a strategy for shortening tuberculosis (TB) treatment by killing non-replicating Mycobacterium tuberculosis within granulomas where the pathogen relies on ...Targeting β-oxidation has been proposed as a strategy for shortening tuberculosis (TB) treatment by killing non-replicating Mycobacterium tuberculosis within granulomas where the pathogen relies on host-derived lipids. The protein EtfD is thought to couple β-oxidation of fatty acids with the respiratory chain in mycobacteria. However, the structure of EtfD is not known and, as the presumed link between two complex processes, its activity has been difficult to measure, impeding its exploitation as a drug target. Here we show that Mycobacterium smegmatis, a fast growing and nonpathogenic model for M. tuberculosis, relies on EtfD for extracting energy from β-oxidation. The electron cryomicroscopy structure of M. smegmatis EtfD reveals an unusual linear [3Fe-4S] cluster that has not been seen in other protein structures, and suggests how EtfD transfers electrons from β-oxidation to the respiratory chain. We devised an assay that couples EtfD activity to a fluorescent readout of proton pumping by the respiratory chain, which can be used to identify compounds that block mycobacteria from using β-oxidation to power oxidative phosphorylation. | |||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  9ojn.cif.gz 9ojn.cif.gz | 151.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb9ojn.ent.gz pdb9ojn.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  9ojn.json.gz 9ojn.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/oj/9ojn https://data.pdbj.org/pub/pdb/validation_reports/oj/9ojn ftp://data.pdbj.org/pub/pdb/validation_reports/oj/9ojn ftp://data.pdbj.org/pub/pdb/validation_reports/oj/9ojn | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  70545MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein / Sugars , 2 types, 2 molecules A

| #1: Protein | Mass: 111045.625 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)References: UniProt: I7FE09 |
|---|---|
| #2: Sugar | ChemComp-LMT / |
-Non-polymers , 5 types, 6 molecules 





| #3: Chemical | ChemComp-MQ9 / | ||||
|---|---|---|---|---|---|
| #4: Chemical | ChemComp-HEM / | ||||
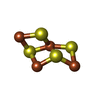
| #5: Chemical | | #6: Chemical | ChemComp-9S8 / | #7: Chemical | ChemComp-A1CBX / | Mass: 295.795 Da / Num. of mol.: 1 / Source method: obtained synthetically / Formula: Fe3S4 / Feature type: SUBJECT OF INVESTIGATION |
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | N |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Structure of Mycobacterium smegmatis EtfD / Type: COMPLEX / Entity ID: #1 / Source: NATURAL |
|---|---|
| Molecular weight | Experimental value: NO |
| Source (natural) | Organism:  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria) |
| Buffer solution | pH: 7.4 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: TFS KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2800 nm / Nominal defocus min: 400 nm |
| Image recording | Electron dose: 70 e/Å2 / Film or detector model: TFS FALCON 4i (4k x 4k) |
- Processing
Processing
| EM software | Name: PHENIX / Version: 1.19.2_4158 / Category: model refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.2 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 48053 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refinement | Highest resolution: 3.2 Å Stereochemistry target values: REAL-SPACE (WEIGHTED MAP SUM AT ATOM CENTERS) | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller




 PDBj
PDBj












