[English] 日本語
 Yorodumi
Yorodumi- PDB-9c1p: Structure of Calcium-Sensing Receptor in complex with positive al... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 9c1p | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of Calcium-Sensing Receptor in complex with positive allosteric modulator '6218 | |||||||||||||||||||||
 Components Components | (Extracellular calcium-sensing ...) x 2 | |||||||||||||||||||||
 Keywords Keywords | MEMBRANE PROTEIN / G-protein coupled receptor / calcium-sensing | |||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of presynaptic membrane potential / bile acid secretion / chemosensory behavior / response to fibroblast growth factor / cellular response to peptide / cellular response to vitamin D / Class C/3 (Metabotropic glutamate/pheromone receptors) / positive regulation of positive chemotaxis / fat pad development / cellular response to hepatocyte growth factor stimulus ...regulation of presynaptic membrane potential / bile acid secretion / chemosensory behavior / response to fibroblast growth factor / cellular response to peptide / cellular response to vitamin D / Class C/3 (Metabotropic glutamate/pheromone receptors) / positive regulation of positive chemotaxis / fat pad development / cellular response to hepatocyte growth factor stimulus / amino acid binding / branching morphogenesis of an epithelial tube / positive regulation of calcium ion import / positive regulation of NLRP3 inflammasome complex assembly / regulation of calcium ion transport / positive regulation of vasoconstriction / anatomical structure morphogenesis / cellular response to low-density lipoprotein particle stimulus / detection of calcium ion / JNK cascade / ossification / axon terminus / response to ischemia / chloride transmembrane transport / cellular response to glucose stimulus / integrin binding / G protein-coupled receptor activity / vasodilation / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / intracellular calcium ion homeostasis / positive regulation of insulin secretion / presynaptic membrane / cellular response to hypoxia / G alpha (i) signalling events / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (q) signalling events / basolateral plasma membrane / transmembrane transporter binding / positive regulation of ERK1 and ERK2 cascade / apical plasma membrane / G protein-coupled receptor signaling pathway / neuronal cell body / calcium ion binding / positive regulation of cell population proliferation / positive regulation of gene expression / protein kinase binding / glutamatergic synapse / cell surface / protein homodimerization activity / identical protein binding / plasma membrane Similarity search - Function | |||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.8 Å | |||||||||||||||||||||
 Authors Authors | Wu, C. / Skiniotis, G. | |||||||||||||||||||||
| Funding support |  United States, 2items United States, 2items
| |||||||||||||||||||||
 Citation Citation |  Journal: Science / Year: 2024 Journal: Science / Year: 2024Title: Large library docking identifies positive allosteric modulators of the calcium-sensing receptor. Authors: Fangyu Liu / Cheng-Guo Wu / Chia-Ling Tu / Isabella Glenn / Justin Meyerowitz / Anat Levit Kaplan / Jiankun Lyu / Zhiqiang Cheng / Olga O Tarkhanova / Yurii S Moroz / John J Irwin / Wenhan ...Authors: Fangyu Liu / Cheng-Guo Wu / Chia-Ling Tu / Isabella Glenn / Justin Meyerowitz / Anat Levit Kaplan / Jiankun Lyu / Zhiqiang Cheng / Olga O Tarkhanova / Yurii S Moroz / John J Irwin / Wenhan Chang / Brian K Shoichet / Georgios Skiniotis /   Abstract: Positive allosteric modulator (PAM) drugs enhance the activation of the calcium-sensing receptor (CaSR) and suppress parathyroid hormone (PTH) secretion. Unfortunately, these hyperparathyroidism- ...Positive allosteric modulator (PAM) drugs enhance the activation of the calcium-sensing receptor (CaSR) and suppress parathyroid hormone (PTH) secretion. Unfortunately, these hyperparathyroidism-treating drugs can induce hypocalcemia and arrhythmias. Seeking improved modulators, we docked libraries of 2.7 million and 1.2 billion molecules against the CaSR structure. The billion-molecule docking found PAMs with a 2.7-fold higher hit rate than the million-molecule library, with hits up to 37-fold more potent. Structure-based optimization led to nanomolar leads. In ex vivo organ assays, one of these PAMs was 100-fold more potent than the standard of care, cinacalcet, and reduced serum PTH levels in mice without the hypocalcemia typical of CaSR drugs. As determined from cryo-electron microscopy structures, the PAMs identified here promote CaSR conformations that more closely resemble the activated state than those induced by the established drugs. | |||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  9c1p.cif.gz 9c1p.cif.gz | 296.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb9c1p.ent.gz pdb9c1p.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  9c1p.json.gz 9c1p.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/c1/9c1p https://data.pdbj.org/pub/pdb/validation_reports/c1/9c1p ftp://data.pdbj.org/pub/pdb/validation_reports/c1/9c1p ftp://data.pdbj.org/pub/pdb/validation_reports/c1/9c1p | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  45127MC  9c2fC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Extracellular calcium-sensing ... , 2 types, 2 molecules AB
| #1: Protein | Mass: 108023.055 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CASR, GPRC2A, PCAR1 / Production host: Homo sapiens (human) / Gene: CASR, GPRC2A, PCAR1 / Production host:  |
|---|---|
| #2: Protein | Mass: 105938.789 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CASR, GPRC2A, PCAR1 / Production host: Homo sapiens (human) / Gene: CASR, GPRC2A, PCAR1 / Production host:  |
-Sugars , 1 types, 6 molecules 
| #3: Sugar | ChemComp-NAG / |
|---|
-Non-polymers , 4 types, 14 molecules 
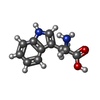



| #4: Chemical | ChemComp-CA / #5: Chemical | #6: Chemical | #7: Chemical | Mass: 337.482 Da / Num. of mol.: 2 / Source method: obtained synthetically / Formula: C20H23N3S / Feature type: SUBJECT OF INVESTIGATION |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Calcium-sensing receptor bound to compound '6218 / Type: COMPLEX / Entity ID: #1-#2 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  |
| Buffer solution | pH: 7.5 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 277 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: TFS KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 1500 nm / Nominal defocus min: 500 nm |
| Image recording | Electron dose: 55 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| EM software | Name: PHENIX / Category: model refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 2.8 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 346741 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller









 PDBj
PDBj






