[English] 日本語
 Yorodumi
Yorodumi- PDB-8gm5: Functional construct of the Eukaryotic elongation factor 2 kinase... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8gm5 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Functional construct of the Eukaryotic elongation factor 2 kinase bound to Calmodulin, ADP and to the A-484954 inhibitor and showing two conformations for the 498-520 loop | ||||||
 Components Components |
| ||||||
 Keywords Keywords | TRANSLATION/Transferase / elongation factor 2 kinase / eEF2 / eEF2K / eEF-2K / Calmodulin / TRANSLATION / A-484954 / ADP / allostery / TRANSLATION-Transferase complex / alternate conformations / SIGNALING PROTEIN | ||||||
| Function / homology |  Function and homology information Function and homology informationelongation factor 2 kinase / elongation factor-2 kinase activity / regulation of translation at postsynapse / response to prolactin / myosin II filament disassembly / cellular response to anoxia / translation factor activity, RNA binding / positive regulation of dendritic spine morphogenesis / positive regulation of synapse assembly / CaM pathway ...elongation factor 2 kinase / elongation factor-2 kinase activity / regulation of translation at postsynapse / response to prolactin / myosin II filament disassembly / cellular response to anoxia / translation factor activity, RNA binding / positive regulation of dendritic spine morphogenesis / positive regulation of synapse assembly / CaM pathway / Cam-PDE 1 activation / Sodium/Calcium exchangers / Calmodulin induced events / Reduction of cytosolic Ca++ levels / Activation of Ca-permeable Kainate Receptor / CREB1 phosphorylation through the activation of CaMKII/CaMKK/CaMKIV cascasde / Loss of phosphorylation of MECP2 at T308 / CREB1 phosphorylation through the activation of Adenylate Cyclase / CaMK IV-mediated phosphorylation of CREB / PKA activation / negative regulation of high voltage-gated calcium channel activity / Glycogen breakdown (glycogenolysis) / CLEC7A (Dectin-1) induces NFAT activation / Activation of RAC1 downstream of NMDARs / negative regulation of ryanodine-sensitive calcium-release channel activity / organelle localization by membrane tethering / mitochondrion-endoplasmic reticulum membrane tethering / autophagosome membrane docking / negative regulation of calcium ion export across plasma membrane / regulation of cardiac muscle cell action potential / presynaptic endocytosis / Synthesis of IP3 and IP4 in the cytosol / regulation of cell communication by electrical coupling involved in cardiac conduction / Phase 0 - rapid depolarisation / calcineurin-mediated signaling / Negative regulation of NMDA receptor-mediated neuronal transmission / Unblocking of NMDA receptors, glutamate binding and activation / RHO GTPases activate PAKs / mTORC1-mediated signalling / Ion transport by P-type ATPases / Uptake and function of anthrax toxins / regulation of ryanodine-sensitive calcium-release channel activity / Long-term potentiation / protein phosphatase activator activity / positive regulation of endocytosis / Calcineurin activates NFAT / Regulation of MECP2 expression and activity / translational elongation / DARPP-32 events / Smooth Muscle Contraction / detection of calcium ion / regulation of cardiac muscle contraction / catalytic complex / RHO GTPases activate IQGAPs / regulation of cardiac muscle contraction by regulation of the release of sequestered calcium ion / cellular response to interferon-beta / Protein methylation / calcium channel inhibitor activity / Activation of AMPK downstream of NMDARs / presynaptic cytosol / regulation of release of sequestered calcium ion into cytosol by sarcoplasmic reticulum / Ion homeostasis / eNOS activation / cellular response to brain-derived neurotrophic factor stimulus / titin binding / Tetrahydrobiopterin (BH4) synthesis, recycling, salvage and regulation / sperm midpiece / regulation of calcium-mediated signaling / voltage-gated potassium channel complex / calcium channel complex / FCERI mediated Ca+2 mobilization / substantia nigra development / regulation of heart rate / Ras activation upon Ca2+ influx through NMDA receptor / FCGR3A-mediated IL10 synthesis / cellular response to calcium ion / Antigen activates B Cell Receptor (BCR) leading to generation of second messengers / calyx of Held / cellular response to cAMP / adenylate cyclase activator activity / sarcomere / response to ischemia / VEGFR2 mediated cell proliferation / protein serine/threonine kinase activator activity / VEGFR2 mediated vascular permeability / regulation of cytokinesis / spindle microtubule / Translocation of SLC2A4 (GLUT4) to the plasma membrane / positive regulation of receptor signaling pathway via JAK-STAT / calcium channel regulator activity / RAF activation / Transcriptional activation of mitochondrial biogenesis / response to calcium ion / cellular response to type II interferon / Stimuli-sensing channels / G2/M transition of mitotic cell cycle / long-term synaptic potentiation / cellular response to insulin stimulus / spindle pole / RAS processing Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.12 Å MOLECULAR REPLACEMENT / Resolution: 2.12 Å | ||||||
 Authors Authors | Piserchio, A. / Isiorho, E.A. / Dalby, K.N. / Ghose, R. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2023 Journal: J.Biol.Chem. / Year: 2023Title: Structure of the complex between calmodulin and a functional construct of eukaryotic elongation factor 2 kinase bound to an ATP-competitive inhibitor. Authors: Piserchio, A. / Isiorho, E.A. / Dalby, K.N. / Ghose, R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8gm5.cif.gz 8gm5.cif.gz | 139.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8gm5.ent.gz pdb8gm5.ent.gz | 101 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8gm5.json.gz 8gm5.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  8gm5_validation.pdf.gz 8gm5_validation.pdf.gz | 1.1 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  8gm5_full_validation.pdf.gz 8gm5_full_validation.pdf.gz | 1.1 MB | Display | |
| Data in XML |  8gm5_validation.xml.gz 8gm5_validation.xml.gz | 23.7 KB | Display | |
| Data in CIF |  8gm5_validation.cif.gz 8gm5_validation.cif.gz | 33.7 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/gm/8gm5 https://data.pdbj.org/pub/pdb/validation_reports/gm/8gm5 ftp://data.pdbj.org/pub/pdb/validation_reports/gm/8gm5 ftp://data.pdbj.org/pub/pdb/validation_reports/gm/8gm5 | HTTPS FTP |
-Related structure data
| Related structure data |  8gm4C  7shqS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||
| Unit cell |
|
- Components
Components
-Protein , 2 types, 2 molecules AB
| #1: Protein | Mass: 60380.727 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: EEF2K / Production host: Homo sapiens (human) / Gene: EEF2K / Production host:  |
|---|---|
| #2: Protein | Mass: 16721.350 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CALM1, CALM, CAM, CAM1 / Production host: Homo sapiens (human) / Gene: CALM1, CALM, CAM, CAM1 / Production host:  |
-Non-polymers , 5 types, 245 molecules 
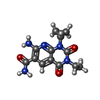







| #3: Chemical | ChemComp-ADP / | ||
|---|---|---|---|
| #4: Chemical | ChemComp-EKI / | ||
| #5: Chemical | ChemComp-ZN / | ||
| #6: Chemical | | #7: Water | ChemComp-HOH / | |
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.68 Å3/Da / Density % sol: 53.71 % |
|---|---|
| Crystal grow | Temperature: 295.15 K / Method: vapor diffusion / pH: 6.5 Details: Cocktail:16.55% PEG-3350, 0.2 M NaF, 100 mM BisTris-Propane Protein solution: 10.3 mg/mL 20 mM Tris pH 7.5, 100 mM NaCl, 3 mM CaCl2, 1mM TCEP, 1.5 m Inhibitor , 3.1 % DMSO 2protein/1cocktail (0.2 ul total) Temp details: Room temperature, not controlled |
-Data collection
| Diffraction | Mean temperature: 100 K / Ambient temp details: Cold nitrogen stream / Serial crystal experiment: N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  NSLS-II NSLS-II  / Beamline: 17-ID-2 / Wavelength: 0.97934 Å / Beamline: 17-ID-2 / Wavelength: 0.97934 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: DECTRIS EIGER X 16M / Detector: PIXEL / Date: May 28, 2022 Details: Horizontal pre-focus bimorph mirror & KB bimorph mirrors | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Monochromator: Si(111) DCM / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.97934 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.12→82.906 Å / Num. obs: 30789 / % possible obs: 92 % / Redundancy: 4.93 % / CC1/2: 0.996 / Rmerge(I) obs: 0.1221 / Rpim(I) all: 0.0603 / Rrim(I) all: 0.1367 / Net I/σ(I): 5.89 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell |
|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 7SHQ Resolution: 2.12→45.94 Å / SU ML: 0.241 / Cross valid method: FREE R-VALUE / σ(F): 1.36 / Phase error: 27.122 Stereochemistry target values: GEOSTD + MONOMER LIBRARY + CDL V1.2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 42.27 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.12→45.94 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller


 PDBj
PDBj

























