+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8df0 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Abp1D receptor binding domain | |||||||||
 Components Components | Abp1D Receptor Binding Domain | |||||||||
 Keywords Keywords | CELL ADHESION / Chaperone usher pathway adhesin receptor binding domain | |||||||||
| Function / homology |  Function and homology information Function and homology information | |||||||||
| Biological species |  Acinetobacter baumannii (bacteria) Acinetobacter baumannii (bacteria) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.1 Å MOLECULAR REPLACEMENT / Resolution: 2.1 Å | |||||||||
 Authors Authors | Tamadonfar, K.O. / Pinkner, J.S. / Dodson, K.W. / Hultgren, S.J. | |||||||||
| Funding support |  United States, 2items United States, 2items
| |||||||||
 Citation Citation |  Journal: Proc.Natl.Acad.Sci.USA / Year: 2023 Journal: Proc.Natl.Acad.Sci.USA / Year: 2023Title: Structure-function correlates of fibrinogen binding by Acinetobacter adhesins critical in catheter-associated urinary tract infections. Authors: Tamadonfar, K.O. / Di Venanzio, G. / Pinkner, J.S. / Dodson, K.W. / Kalas, V. / Zimmerman, M.I. / Bazan Villicana, J. / Bowman, G.R. / Feldman, M.F. / Hultgren, S.J. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8df0.cif.gz 8df0.cif.gz | 191.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8df0.ent.gz pdb8df0.ent.gz | 156.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8df0.json.gz 8df0.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/df/8df0 https://data.pdbj.org/pub/pdb/validation_reports/df/8df0 ftp://data.pdbj.org/pub/pdb/validation_reports/df/8df0 ftp://data.pdbj.org/pub/pdb/validation_reports/df/8df0 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  8dezSC 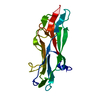 8dkaC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 18846.119 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Acinetobacter baumannii (bacteria) Acinetobacter baumannii (bacteria)Gene: fimA, AYR68_10060, B7L45_11295, BAA1790NC_1605, BS065_10665, CBL15_10290, CTZ19_10795, D8O08_010130, DLI72_16175, E2532_11475, E2533_08890, E2534_05850, E2535_07595, E2536_00985, E2538_06840, ...Gene: fimA, AYR68_10060, B7L45_11295, BAA1790NC_1605, BS065_10665, CBL15_10290, CTZ19_10795, D8O08_010130, DLI72_16175, E2532_11475, E2533_08890, E2534_05850, E2535_07595, E2536_00985, E2538_06840, E2539_15695, E2540_01165, E2541_05705, EA720_002470, FE003_11010, H1058_10155, HIN86_11385, IAG11_17165, SAMEA104305340_00065 Production host:  #2: Water | ChemComp-HOH / | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 4.96 Å3/Da / Density % sol: 75.21 % |
|---|---|
| Crystal grow | Temperature: 292.15 K / Method: vapor diffusion, hanging drop / Details: 0.8 M (NH4)2SO4, 0.025 M Citric Acid pH 3.56 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ALS ALS  / Beamline: 4.2.2 / Wavelength: 0.9762 Å / Beamline: 4.2.2 / Wavelength: 0.9762 Å |
| Detector | Type: RDI CMOS_8M / Detector: CMOS / Date: Oct 25, 2019 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9762 Å / Relative weight: 1 |
| Reflection | Resolution: 2.1→44.51 Å / Num. obs: 45137 / % possible obs: 99.76 % / Redundancy: 13.9 % / Biso Wilson estimate: 33.97 Å2 / CC1/2: 0.996 / CC star: 0.999 / Rmerge(I) obs: 0.2715 / Rpim(I) all: 0.07507 / Rrim(I) all: 0.2818 / Net I/σ(I): 11.9 |
| Reflection shell | Resolution: 2.1→2.175 Å / Redundancy: 14 % / Rmerge(I) obs: 2.938 / Num. unique obs: 4370 / CC1/2: 0.434 / CC star: 0.778 / Rpim(I) all: 0.8113 / Rrim(I) all: 3.049 / % possible all: 99.36 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 8DEZ Resolution: 2.1→44.51 Å / SU ML: 0.25 / Cross valid method: THROUGHOUT / σ(F): 1.33 / Phase error: 23.8 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 93.76 Å2 / Biso mean: 42.8553 Å2 / Biso min: 23.93 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.1→44.51 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: -5.6049 Å / Origin y: 21.9195 Å / Origin z: -22.4341 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller



 PDBj
PDBj
