[English] 日本語
 Yorodumi
Yorodumi- PDB-8byn: Chronic traumatic encephalopathy tau filaments with PET ligand fl... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8byn | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
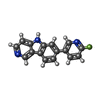
| Title | Chronic traumatic encephalopathy tau filaments with PET ligand flortaucipir | |||||||||||||||||||||
 Components Components | Microtubule-associated protein tau | |||||||||||||||||||||
 Keywords Keywords | PROTEIN FIBRIL / Chronic traumatic encephalopathy / Primary age-related tauopathy / Tau filaments / PET ligand / Flortaucipir | |||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationplus-end-directed organelle transport along microtubule / histone-dependent DNA binding / negative regulation of protein localization to mitochondrion / neurofibrillary tangle / microtubule lateral binding / axonal transport / tubulin complex / positive regulation of protein localization to synapse / phosphatidylinositol bisphosphate binding / generation of neurons ...plus-end-directed organelle transport along microtubule / histone-dependent DNA binding / negative regulation of protein localization to mitochondrion / neurofibrillary tangle / microtubule lateral binding / axonal transport / tubulin complex / positive regulation of protein localization to synapse / phosphatidylinositol bisphosphate binding / generation of neurons / rRNA metabolic process / axonal transport of mitochondrion / regulation of mitochondrial fission / axon development / regulation of microtubule-based movement / intracellular distribution of mitochondria / regulation of chromosome organization / central nervous system neuron development / minor groove of adenine-thymine-rich DNA binding / lipoprotein particle binding / microtubule polymerization / negative regulation of mitochondrial membrane potential / regulation of microtubule polymerization / dynactin binding / apolipoprotein binding / protein polymerization / main axon / axolemma / Caspase-mediated cleavage of cytoskeletal proteins / regulation of microtubule polymerization or depolymerization / negative regulation of mitochondrial fission / glial cell projection / neurofibrillary tangle assembly / positive regulation of axon extension / positive regulation of microtubule polymerization / regulation of cellular response to heat / Activation of AMPK downstream of NMDARs / positive regulation of protein localization / positive regulation of superoxide anion generation / supramolecular fiber organization / cellular response to brain-derived neurotrophic factor stimulus / regulation of long-term synaptic depression / cytoplasmic microtubule organization / regulation of calcium-mediated signaling / somatodendritic compartment / axon cytoplasm / synapse assembly / astrocyte activation / phosphatidylinositol binding / nuclear periphery / enzyme inhibitor activity / protein phosphatase 2A binding / stress granule assembly / regulation of microtubule cytoskeleton organization / regulation of autophagy / cellular response to reactive oxygen species / microglial cell activation / cellular response to nerve growth factor stimulus / Hsp90 protein binding / protein homooligomerization / PKR-mediated signaling / SH3 domain binding / synapse organization / regulation of synaptic plasticity / response to lead ion / microtubule cytoskeleton organization / memory / neuron projection development / cytoplasmic ribonucleoprotein granule / cell-cell signaling / single-stranded DNA binding / cellular response to heat / growth cone / protein-folding chaperone binding / microtubule cytoskeleton / actin binding / cell body / double-stranded DNA binding / microtubule binding / sequence-specific DNA binding / amyloid fibril formation / dendritic spine / microtubule / protein-macromolecule adaptor activity / learning or memory / neuron projection / membrane raft / negative regulation of gene expression / axon / neuronal cell body / DNA damage response / dendrite / protein kinase binding / enzyme binding / mitochondrion / DNA binding / RNA binding / extracellular region / identical protein binding / nucleus Similarity search - Function | |||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / helical reconstruction / cryo EM / Resolution: 2.6 Å | |||||||||||||||||||||
 Authors Authors | Shi, Y. / Ghetti, B. / Goedert, M. / Scheres, S.H.W. | |||||||||||||||||||||
| Funding support |  United Kingdom, United Kingdom,  Switzerland, Switzerland,  United States, 6items United States, 6items
| |||||||||||||||||||||
 Citation Citation |  Journal: J Mol Biol / Year: 2023 Journal: J Mol Biol / Year: 2023Title: Cryo-EM Structures of Chronic Traumatic Encephalopathy Tau Filaments with PET Ligand Flortaucipir. Authors: Yang Shi / Bernardino Ghetti / Michel Goedert / Sjors H W Scheres /   Abstract: Positron emission tomography (PET) imaging allows monitoring the progression of amyloid aggregation in the living brain. [F]-Flortaucipir is the only approved PET tracer compound for the ...Positron emission tomography (PET) imaging allows monitoring the progression of amyloid aggregation in the living brain. [F]-Flortaucipir is the only approved PET tracer compound for the visualisation of tau aggregation. Here, we describe cryo-EM experiments on tau filaments in the presence and absence of flortaucipir. We used tau filaments isolated from the brain of an individual with Alzheimer's disease (AD), and from the brain of an individual with primary age-related tauopathy (PART) with a co-pathology of chronic traumatic encephalopathy (CTE). Unexpectedly, we were unable to visualise additional cryo-EM density for flortaucipir for AD paired helical or straight filaments (PHFs or SFs), but we did observe density for flortaucipir binding to CTE Type I filaments from the case with PART. In the latter, flortaucipir binds in a 1:1 molecular stoichiometry with tau, adjacent to lysine 353 and aspartate 358. By adopting a tilted geometry with respect to the helical axis, the 4.7 Å distance between neighbouring tau monomers is reconciled with the 3.5 Å distance consistent with π-π-stacking between neighbouring molecules of flortaucipir. | |||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8byn.cif.gz 8byn.cif.gz | 116.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8byn.ent.gz pdb8byn.ent.gz | 75.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8byn.json.gz 8byn.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/by/8byn https://data.pdbj.org/pub/pdb/validation_reports/by/8byn ftp://data.pdbj.org/pub/pdb/validation_reports/by/8byn ftp://data.pdbj.org/pub/pdb/validation_reports/by/8byn | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  16329MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 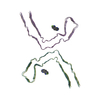
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 45919.871 Da / Num. of mol.: 6 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P10636 Homo sapiens (human) / References: UniProt: P10636#2: Chemical | ChemComp-S9C / Has ligand of interest | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: FILAMENT / 3D reconstruction method: helical reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Sarkosyl-insoluble fraction from the hippocampus of an individual with primary age-related tauopathy Type: TISSUE / Entity ID: #1 / Source: NATURAL |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Buffer solution | pH: 7.4 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2000 nm / Nominal defocus min: 1000 nm |
| Image recording | Electron dose: 40 e/Å2 / Film or detector model: FEI FALCON IV (4k x 4k) |
- Processing
Processing
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
|---|---|
| Helical symmerty | Angular rotation/subunit: 179.43 ° / Axial rise/subunit: 2.37 Å / Axial symmetry: C1 |
| 3D reconstruction | Resolution: 2.6 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 58019 / Symmetry type: HELICAL |
 Movie
Movie Controller
Controller









 PDBj
PDBj