[English] 日本語
 Yorodumi
Yorodumi- PDB-7ypy: Bovine heart cytochrome c oxidase in fully oxidized state at 1.5 ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7ypy | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Bovine heart cytochrome c oxidase in fully oxidized state at 1.5 angstrom resolution | |||||||||
 Components Components | (Cytochrome c oxidase subunit ...) x 14 | |||||||||
 Keywords Keywords | OXIDOREDUCTASE / Respiratory enzyme / Membrane protein / Hemeprotein / Protein complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationComplex IV assembly / TP53 Regulates Metabolic Genes / respiratory chain complex IV assembly / Cytoprotection by HMOX1 / mitochondrial respirasome assembly / Respiratory electron transport / respiratory chain complex IV / respiratory chain complex / Mitochondrial translation termination / cytochrome-c oxidase ...Complex IV assembly / TP53 Regulates Metabolic Genes / respiratory chain complex IV assembly / Cytoprotection by HMOX1 / mitochondrial respirasome assembly / Respiratory electron transport / respiratory chain complex IV / respiratory chain complex / Mitochondrial translation termination / cytochrome-c oxidase / oxidative phosphorylation / mitochondrial electron transport, cytochrome c to oxygen / cytochrome-c oxidase activity / Mitochondrial protein degradation / ATP synthesis coupled electron transport / enzyme regulator activity / proton transmembrane transport / aerobic respiration / respiratory electron transport chain / central nervous system development / mitochondrial membrane / oxidoreductase activity / mitochondrial inner membrane / copper ion binding / heme binding / mitochondrion / metal ion binding Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.5 Å MOLECULAR REPLACEMENT / Resolution: 1.5 Å | |||||||||
 Authors Authors | Shimada, A. / Tsukihara, T. | |||||||||
| Funding support |  Japan, 2items Japan, 2items
| |||||||||
 Citation Citation |  Journal: J. Biol. Chem. / Year: 2016 Journal: J. Biol. Chem. / Year: 2016Title: The Mg2+-containing water cluster of mammalian cytochrome c oxidase collects four pumping proton equivalents in each catalytic cycle. Authors: Yano, N. / Muramoto, K. / Shimada, A. / Shinzawa-Itoh, K. / Tsukihara, T. / Yoshikawa, S. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7ypy.cif.gz 7ypy.cif.gz | 1.5 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7ypy.ent.gz pdb7ypy.ent.gz | 1.3 MB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7ypy.json.gz 7ypy.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/yp/7ypy https://data.pdbj.org/pub/pdb/validation_reports/yp/7ypy ftp://data.pdbj.org/pub/pdb/validation_reports/yp/7ypy ftp://data.pdbj.org/pub/pdb/validation_reports/yp/7ypy | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5b1aSC  5b1bC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Cytochrome c oxidase subunit ... , 14 types, 26 molecules ANBOCPDQERFSGHUIVJWKXLYMZT
| #1: Protein | Mass: 57093.852 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #2: Protein | Mass: 26068.404 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #3: Protein | Mass: 29957.627 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #4: Protein | Mass: 17179.646 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #5: Protein | Mass: 12453.081 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #6: Protein | Mass: 10684.038 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #7: Protein | | Mass: 9629.782 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  #8: Protein | Mass: 10039.244 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #9: Protein | Mass: 8521.021 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #10: Protein | Mass: 6682.726 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #11: Protein | Mass: 6365.217 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #12: Protein/peptide | Mass: 5449.396 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #13: Protein/peptide | Mass: 4967.756 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #14: Protein | | Mass: 9549.802 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|
-Sugars , 1 types, 26 molecules 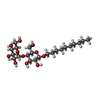
| #23: Sugar | ChemComp-DMU / |
|---|
-Non-polymers , 16 types, 2945 molecules 








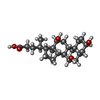





















| #15: Chemical | ChemComp-HEA / #16: Chemical | #17: Chemical | #18: Chemical | #19: Chemical | #20: Chemical | ChemComp-PGV / ( #21: Chemical | ChemComp-TGL / #22: Chemical | ChemComp-EDO / #24: Chemical | #25: Chemical | ChemComp-CHD / #26: Chemical | #27: Chemical | ChemComp-CDL / #28: Chemical | ChemComp-PEK / ( #29: Chemical | #30: Chemical | #31: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 4.03 Å3/Da / Density % sol: 69.49 % / Description: tetragonal plate |
|---|---|
| Crystal grow | Temperature: 277 K / Method: batch mode / pH: 6.8 / Details: Sodium phosphate buffer, PEG 4000 |
-Data collection
| Diffraction | Mean temperature: 50 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SPring-8 SPring-8  / Beamline: BL44XU / Wavelength: 0.9 Å / Beamline: BL44XU / Wavelength: 0.9 Å |
| Detector | Type: RAYONIX MX225HE / Detector: CCD / Date: Apr 23, 2010 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9 Å / Relative weight: 1 |
| Reflection | Resolution: 1.5→200 Å / Num. obs: 1041861 / % possible obs: 100 % / Redundancy: 14.9 % / Rmerge(I) obs: 0.107 / Net I/σ(I): 32.7 |
| Reflection shell | Resolution: 1.5→1.52 Å / Redundancy: 14.8 % / Mean I/σ(I) obs: 3.9 / Num. unique obs: 34593 / Rpim(I) all: 0.296 / % possible all: 100 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5B1A Resolution: 1.5→39.955 Å / SU ML: 0.11 / Cross valid method: FREE R-VALUE / σ(F): 2 / Phase error: 19.42 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.5→39.955 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller


 PDBj
PDBj



















