+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7u1b | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of EstG in complex with tantalum cluster | ||||||
 Components Components | Beta-lactamase domain-containing protein | ||||||
 Keywords Keywords | HYDROLASE / alpha/beta fold / CCNA_01638 | ||||||
| Function / homology | : / Beta-lactamase-related / Beta-lactamase / Beta-lactamase/transpeptidase-like / HEXATANTALUM DODECABROMIDE / Beta-lactamase-related domain-containing protein Function and homology information Function and homology information | ||||||
| Biological species |  Caulobacter vibrioides (bacteria) Caulobacter vibrioides (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.62 Å MOLECULAR REPLACEMENT / Resolution: 2.62 Å | ||||||
 Authors Authors | Chen, Z. / Gabelli, S.B. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Curr.Biol. / Year: 2023 Journal: Curr.Biol. / Year: 2023Title: EstG is a novel esterase required for cell envelope integrity in Caulobacter. Authors: Daitch, A.K. / Orsburn, B.C. / Chen, Z. / Alvarez, L. / Eberhard, C.D. / Sundararajan, K. / Zeinert, R. / Kreitler, D.F. / Jakoncic, J. / Chien, P. / Cava, F. / Gabelli, S.B. / Goley, E.D. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7u1b.cif.gz 7u1b.cif.gz | 92.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7u1b.ent.gz pdb7u1b.ent.gz | 67.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7u1b.json.gz 7u1b.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/u1/7u1b https://data.pdbj.org/pub/pdb/validation_reports/u1/7u1b ftp://data.pdbj.org/pub/pdb/validation_reports/u1/7u1b ftp://data.pdbj.org/pub/pdb/validation_reports/u1/7u1b | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7u1cC  7udaC  1ci8S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 49922.344 Da / Num. of mol.: 1 Fragment: estG, Esterase for Stress Tolerance acting on glucans Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Caulobacter vibrioides (bacteria) / Strain: ATCC 19089 / CB15 / Gene: CC_1567 / Plasmid: pEG1622 / Production host: Caulobacter vibrioides (bacteria) / Strain: ATCC 19089 / CB15 / Gene: CC_1567 / Plasmid: pEG1622 / Production host:  |
|---|
-Non-polymers , 5 types, 14 molecules 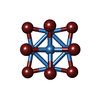







| #2: Chemical | ChemComp-TBR / |
|---|---|
| #3: Chemical | ChemComp-BCN / |
| #4: Chemical | ChemComp-TRS / |
| #5: Chemical | ChemComp-SO4 / |
| #6: Water | ChemComp-HOH / |
-Details
| Has ligand of interest | N |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.64 Å3/Da / Density % sol: 66.21 % / Mosaicity: 0.13 ° |
|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion / pH: 8.5 Details: 20% PEG500 MME, 10% PEG20000, 0.1 M Tris/Bicine pH 8.5 and 90 mM mixture of sodium nitrate, sodium phosphate dibasic and ammonium sulfate |
-Data collection
| Diffraction | Mean temperature: 277 K / Serial crystal experiment: N | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Type: OTHER / Wavelength: 0.92009 Å SYNCHROTRON / Type: OTHER / Wavelength: 0.92009 Å | ||||||||||||||||||||||||||||||
| Detector | Type: DECTRIS EIGER X 9M / Detector: PIXEL / Date: Jul 16, 2021 | ||||||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.92009 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.62→28.71 Å / Num. obs: 21379 / % possible obs: 99.5 % / Redundancy: 9.1 % / CC1/2: 0.998 / Rmerge(I) obs: 0.107 / Rpim(I) all: 0.037 / Rrim(I) all: 0.113 / Net I/σ(I): 16.9 / Num. measured all: 195511 | ||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1CI8 Resolution: 2.62→27.96 Å / Cor.coef. Fo:Fc: 0.935 / Cor.coef. Fo:Fc free: 0.913 / WRfactor Rfree: 0.2133 / WRfactor Rwork: 0.1755 / FOM work R set: 0.8299 / SU B: 8.745 / SU ML: 0.177 / SU R Cruickshank DPI: 0.3214 / SU Rfree: 0.2455 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.321 / ESU R Free: 0.246 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS U VALUES : REFINED INDIVIDUALLY
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 420.63 Å2 / Biso mean: 49.622 Å2 / Biso min: 25.32 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.62→27.96 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.625→2.693 Å / Rfactor Rfree error: 0 / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller



 PDBj
PDBj





