+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: PDB / ID: 7tzo | ||||||
|---|---|---|---|---|---|---|---|
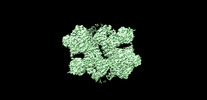
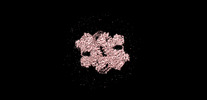
| タイトル | The apo structure of human mTORC2 complex | ||||||
 要素 要素 |
| ||||||
 キーワード キーワード | SIGNALING PROTEIN / complex | ||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報TORC2 signaling / regulation of peptidyl-serine phosphorylation / positive regulation of cytoplasmic translational initiation / positive regulation of pentose-phosphate shunt / RNA polymerase III type 1 promoter sequence-specific DNA binding / RNA polymerase III type 2 promoter sequence-specific DNA binding / T-helper 1 cell lineage commitment / regulation of locomotor rhythm / positive regulation of wound healing, spreading of epidermal cells / cellular response to leucine starvation ...TORC2 signaling / regulation of peptidyl-serine phosphorylation / positive regulation of cytoplasmic translational initiation / positive regulation of pentose-phosphate shunt / RNA polymerase III type 1 promoter sequence-specific DNA binding / RNA polymerase III type 2 promoter sequence-specific DNA binding / T-helper 1 cell lineage commitment / regulation of locomotor rhythm / positive regulation of wound healing, spreading of epidermal cells / cellular response to leucine starvation / regulation of membrane permeability / TFIIIC-class transcription factor complex binding / heart valve morphogenesis / negative regulation of lysosome organization / RNA polymerase III type 3 promoter sequence-specific DNA binding / TORC2 complex / TORC1 complex / positive regulation of transcription of nucleolar large rRNA by RNA polymerase I / regulation of cellular response to oxidative stress / regulation of autophagosome assembly / calcineurin-NFAT signaling cascade / nucleus localization / TORC1 signaling / voluntary musculoskeletal movement / regulation of osteoclast differentiation / positive regulation of keratinocyte migration / cellular response to L-leucine / MTOR signalling / Amino acids regulate mTORC1 / cellular response to nutrient / energy reserve metabolic process / phosphatidic acid binding / Energy dependent regulation of mTOR by LKB1-AMPK / negative regulation of cell size / ruffle organization / phosphatidylinositol-3,4-bisphosphate binding / negative regulation of Ras protein signal transduction / cellular response to osmotic stress / negative regulation of protein localization to nucleus / anoikis / cardiac muscle cell development / phosphatidylinositol-3,5-bisphosphate binding / regulation of establishment of cell polarity / embryo development ending in birth or egg hatching / positive regulation of transcription by RNA polymerase III / negative regulation of calcineurin-NFAT signaling cascade / regulation of myelination / regulation of cell size / Macroautophagy / positive regulation of oligodendrocyte differentiation / negative regulation of macroautophagy / positive regulation of actin filament polymerization / lysosome organization / positive regulation of myotube differentiation / behavioral response to pain / oligodendrocyte differentiation / regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / mTORC1-mediated signalling / Constitutive Signaling by AKT1 E17K in Cancer / germ cell development / CD28 dependent PI3K/Akt signaling / cellular response to nutrient levels / phosphatidylinositol-3,4,5-trisphosphate binding / positive regulation of phosphoprotein phosphatase activity / HSF1-dependent transactivation / positive regulation of TOR signaling / TOR signaling / neuronal action potential / positive regulation of translational initiation / response to amino acid / regulation of macroautophagy / endomembrane system / 'de novo' pyrimidine nucleobase biosynthetic process / positive regulation of lamellipodium assembly / positive regulation of epithelial to mesenchymal transition / positive regulation of lipid biosynthetic process / heart morphogenesis / cardiac muscle contraction / regulation of cellular response to heat / positive regulation of stress fiber assembly / cytoskeleton organization / positive regulation of endothelial cell proliferation / T cell costimulation / substantia nigra development / phosphatidylinositol-4,5-bisphosphate binding / cellular response to amino acid starvation / positive regulation of glycolytic process / cellular response to starvation / phagocytic vesicle / negative regulation of autophagy / protein serine/threonine kinase activator activity / response to nutrient levels / response to nutrient / post-embryonic development / VEGFR2 mediated vascular permeability / regulation of signal transduction by p53 class mediator / Regulation of PTEN gene transcription / positive regulation of translation / regulation of cell growth / regulation of actin cytoskeleton organization 類似検索 - 分子機能 | ||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | ||||||
| 手法 | 電子顕微鏡法 / 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.28 Å | ||||||
 データ登録者 データ登録者 | Yu, Z. / Chen, J. / Pearce, D. | ||||||
| 資金援助 |  米国, 1件 米国, 1件
| ||||||
 引用 引用 |  ジャーナル: J Biol Chem / 年: 2022 ジャーナル: J Biol Chem / 年: 2022タイトル: Interactions between mTORC2 core subunits Rictor and mSin1 dictate selective and context-dependent phosphorylation of substrate kinases SGK1 and Akt. 著者: Zanlin Yu / Junliang Chen / Enzo Takagi / Feng Wang / Bidisha Saha / Xi Liu / Lydia-Marie Joubert / Catherine E Gleason / Mingliang Jin / Chengmin Li / Carlos Nowotny / David Agard / Yifan Cheng / David Pearce /  要旨: Mechanistic target of rapamycin complex 2 (mTORC2) is a multi-subunit kinase complex, central to multiple essential signaling pathways. Two core subunits, Rictor and mSin1, distinguish it from the ...Mechanistic target of rapamycin complex 2 (mTORC2) is a multi-subunit kinase complex, central to multiple essential signaling pathways. Two core subunits, Rictor and mSin1, distinguish it from the related mTORC1 and support context-dependent phosphorylation of its substrates. mTORC2 structures have been determined previously; however, important questions remain, particularly regarding the structural determinants mediating substrate specificity and context-dependent activity. Here, we used cryo-EM to obtain high-resolution structures of the human mTORC2 apo-complex in the presence of substrates Akt and SGK1. Using functional assays, we then tested predictions suggested by substrate-induced structural changes in mTORC2. For the first time, we visualized in the apo-state the side chain interactions between Rictor and mTOR that sterically occlude recruitment of mTORC1 substrates and confer resistance to the mTORC1 inhibitor rapamycin. Also in the apo-state, we observed that mSin1 formed extensive contacts with Rictor via a pair of short α-helices nestled between two Rictor helical repeat clusters, as well as by an extended strand that makes multiple weak contacts with Rictor helical cluster 1. In co-complex structures, we found that SGK1, but not Akt, markedly altered the conformation of the mSin1 N-terminal extended strand, disrupting multiple weak interactions while inducing a large rotation of mSin1 residue Arg-83, which then interacts with a patch of negatively charged residues within Rictor. Finally, we demonstrate mutation of Arg-83 to Ala selectively disrupts mTORC2-dependent phosphorylation of SGK1, but not of Akt, supporting context-dependent substrate selection. These findings provide new structural and functional insights into mTORC2 specificity and context-dependent activity. | ||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 構造ビューア | 分子:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- ダウンロードとリンク
ダウンロードとリンク
- ダウンロード
ダウンロード
| PDBx/mmCIF形式 |  7tzo.cif.gz 7tzo.cif.gz | 1.3 MB | 表示 |  PDBx/mmCIF形式 PDBx/mmCIF形式 |
|---|---|---|---|---|
| PDB形式 |  pdb7tzo.ent.gz pdb7tzo.ent.gz | 1 MB | 表示 |  PDB形式 PDB形式 |
| PDBx/mmJSON形式 |  7tzo.json.gz 7tzo.json.gz | ツリー表示 |  PDBx/mmJSON形式 PDBx/mmJSON形式 | |
| その他 |  その他のダウンロード その他のダウンロード |
-検証レポート
| 文書・要旨 |  7tzo_validation.pdf.gz 7tzo_validation.pdf.gz | 1.2 MB | 表示 |  wwPDB検証レポート wwPDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  7tzo_full_validation.pdf.gz 7tzo_full_validation.pdf.gz | 1.4 MB | 表示 | |
| XML形式データ |  7tzo_validation.xml.gz 7tzo_validation.xml.gz | 198 KB | 表示 | |
| CIF形式データ |  7tzo_validation.cif.gz 7tzo_validation.cif.gz | 296.2 KB | 表示 | |
| アーカイブディレクトリ |  https://data.pdbj.org/pub/pdb/validation_reports/tz/7tzo https://data.pdbj.org/pub/pdb/validation_reports/tz/7tzo ftp://data.pdbj.org/pub/pdb/validation_reports/tz/7tzo ftp://data.pdbj.org/pub/pdb/validation_reports/tz/7tzo | HTTPS FTP |
-関連構造データ
| 関連構造データ |  26213MC M: このデータのモデリングに利用したマップデータ C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
- 集合体
集合体
| 登録構造単位 | 
|
|---|---|
| 1 |
|
- 要素
要素
| #1: タンパク質 | 分子量: 302330.406 Da / 分子数: 2 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: MTOR, FRAP, FRAP1, FRAP2, RAFT1, RAPT1 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: MTOR, FRAP, FRAP1, FRAP2, RAFT1, RAPT1 / 発現宿主:  Homo sapiens (ヒト) Homo sapiens (ヒト)参照: UniProt: P42345, non-specific serine/threonine protein kinase #2: タンパク質 | 分子量: 37998.254 Da / 分子数: 2 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: MLST8, GBL, LST8 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: MLST8, GBL, LST8 / 発現宿主:  Homo sapiens (ヒト) / 参照: UniProt: Q9BVC4 Homo sapiens (ヒト) / 参照: UniProt: Q9BVC4#3: タンパク質 | 分子量: 193846.328 Da / 分子数: 2 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: RICTOR, KIAA1999 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: RICTOR, KIAA1999 / 発現宿主:  Homo sapiens (ヒト) / 参照: UniProt: Q6R327 Homo sapiens (ヒト) / 参照: UniProt: Q6R327#4: タンパク質 | 分子量: 60732.328 Da / 分子数: 2 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: MAPKAP1, MIP1, SIN1 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: MAPKAP1, MIP1, SIN1 / 発現宿主:  Homo sapiens (ヒト) / 参照: UniProt: Q9BPZ7 Homo sapiens (ヒト) / 参照: UniProt: Q9BPZ7 |
|---|
-実験情報
-実験
| 実験 | 手法: 電子顕微鏡法 |
|---|---|
| EM実験 | 試料の集合状態: 3D ARRAY / 3次元再構成法: 単粒子再構成法 |
- 試料調製
試料調製
| 構成要素 | 名称: apostate of mTORC2 complex, composed of mTOR, Rictor, mLST8 and mSin1 タイプ: COMPLEX / Entity ID: all / 由来: RECOMBINANT |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 由来(組換発現) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 緩衝液 | pH: 7.5 |
| 試料 | 濃度: 1 mg/ml / 包埋: NO / シャドウイング: NO / 染色: NO / 凍結: YES |
| 試料支持 | グリッドの材料: GOLD / グリッドのサイズ: 300 divisions/in. / グリッドのタイプ: Homemade |
| 急速凍結 | 装置: FEI VITROBOT MARK IV / 凍結剤: ETHANE / 湿度: 100 % / 凍結前の試料温度: 22 K |
- 電子顕微鏡撮影
電子顕微鏡撮影
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
|---|---|
| 顕微鏡 | モデル: FEI TITAN KRIOS |
| 電子銃 | 電子線源:  FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM |
| 電子レンズ | モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2000 nm / 最小 デフォーカス(公称値): 1000 nm |
| 撮影 | 電子線照射量: 60 e/Å2 / フィルム・検出器のモデル: GATAN K3 (6k x 4k) |
- 解析
解析
| CTF補正 | タイプ: NONE |
|---|---|
| 3次元再構成 | 解像度: 3.28 Å / 解像度の算出法: FSC 0.143 CUT-OFF / 粒子像の数: 288538 / 対称性のタイプ: POINT |
 ムービー
ムービー コントローラー
コントローラー



 PDBj
PDBj










