[English] 日本語
 Yorodumi
Yorodumi- PDB-7r7k: Structure of Human Anaplastic Lymphoma Kinase Domain in complex w... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7r7k | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure of Human Anaplastic Lymphoma Kinase Domain in complex with (4-[6-amino-5-[(1~{R})-1-[5-fluoro-2-(triazol-2-yl)phenyl]ethoxy]-3-pyridyl]isoindolin-1-one) | ||||||
 Components Components | ALK tyrosine kinase receptor | ||||||
 Keywords Keywords | TRANSFERASE / kinase | ||||||
| Function / homology |  Function and homology information Function and homology informationASP-3026-resistant ALK mutants / NVP-TAE684-resistant ALK mutants / alectinib-resistant ALK mutants / brigatinib-resistant ALK mutants / ceritinib-resistant ALK mutants / crizotinib-resistant ALK mutants / lorlatinib-resistant ALK mutants / MDK and PTN in ALK signaling / receptor signaling protein tyrosine kinase activator activity / regulation of dopamine receptor signaling pathway ...ASP-3026-resistant ALK mutants / NVP-TAE684-resistant ALK mutants / alectinib-resistant ALK mutants / brigatinib-resistant ALK mutants / ceritinib-resistant ALK mutants / crizotinib-resistant ALK mutants / lorlatinib-resistant ALK mutants / MDK and PTN in ALK signaling / receptor signaling protein tyrosine kinase activator activity / regulation of dopamine receptor signaling pathway / response to environmental enrichment / ALK mutants bind TKIs / peptidyl-tyrosine autophosphorylation / phosphorylation / positive regulation of dendrite development / regulation of neuron differentiation / Signaling by ALK / adult behavior / response to stress / negative regulation of lipid catabolic process / neuron development / energy homeostasis / swimming behavior / transmembrane receptor protein tyrosine kinase activity / cell surface receptor protein tyrosine kinase signaling pathway / : / hippocampus development / receptor protein-tyrosine kinase / Signaling by ALK fusions and activated point mutants / protein autophosphorylation / regulation of cell population proliferation / heparin binding / protein tyrosine kinase activity / regulation of apoptotic process / signaling receptor complex / signal transduction / protein-containing complex / extracellular exosome / ATP binding / identical protein binding / plasma membrane Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  FOURIER SYNTHESIS / Resolution: 1.831 Å FOURIER SYNTHESIS / Resolution: 1.831 Å | ||||||
 Authors Authors | McTigue, M. | ||||||
 Citation Citation |  Journal: Nat Cancer / Year: 2022 Journal: Nat Cancer / Year: 2022Title: Analysis of lorlatinib analogs reveals a roadmap for targeting diverse compound resistance mutations in ALK-positive lung cancer. Authors: Shiba-Ishii, A. / Johnson, T.W. / Dagogo-Jack, I. / Mino-Kenudson, M. / Johnson, T.R. / Wei, P. / Weinrich, S.L. / McTigue, M.A. / Walcott, M.A. / Nguyen-Phuong, L. / Dionne, K. / Acker, A. ...Authors: Shiba-Ishii, A. / Johnson, T.W. / Dagogo-Jack, I. / Mino-Kenudson, M. / Johnson, T.R. / Wei, P. / Weinrich, S.L. / McTigue, M.A. / Walcott, M.A. / Nguyen-Phuong, L. / Dionne, K. / Acker, A. / Kiedrowski, L.A. / Do, A. / Peterson, J.L. / Barth, J.L. / Yeap, B.Y. / Gainor, J.F. / Lin, J.J. / Yoda, S. / Hata, A.N. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7r7k.cif.gz 7r7k.cif.gz | 80.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7r7k.ent.gz pdb7r7k.ent.gz | 57.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7r7k.json.gz 7r7k.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/r7/7r7k https://data.pdbj.org/pub/pdb/validation_reports/r7/7r7k ftp://data.pdbj.org/pub/pdb/validation_reports/r7/7r7k ftp://data.pdbj.org/pub/pdb/validation_reports/r7/7r7k | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7r7rC  2xp2S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 36909.355 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: ALK / Plasmid: PFASTBAC / Cell (production host): SF9 / Production host: Homo sapiens (human) / Gene: ALK / Plasmid: PFASTBAC / Cell (production host): SF9 / Production host:  References: UniProt: Q9UM73, receptor protein-tyrosine kinase |
|---|---|
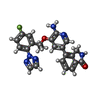
| #2: Chemical | ChemComp-25J / |
| #3: Water | ChemComp-HOH / |
| Has ligand of interest | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.11 Å3/Da / Density % sol: 41.84 % |
|---|---|
| Crystal grow | Temperature: 286 K / Method: vapor diffusion, hanging drop Details: HANGING DROP VAPOR DIFFUSION AT 13 DEGREES C. EQUAL VOLUMES OF PURIFIED PROTEIN SOLUTION(APPROXIMATELY 13-15 MG/ML)CONTAINING 0.0011M INHIBITOR COMPOUND WERE COMBINED WITH A SOLUTION ...Details: HANGING DROP VAPOR DIFFUSION AT 13 DEGREES C. EQUAL VOLUMES OF PURIFIED PROTEIN SOLUTION(APPROXIMATELY 13-15 MG/ML)CONTAINING 0.0011M INHIBITOR COMPOUND WERE COMBINED WITH A SOLUTION CONTAINING: 0.15M AMMONIUM SULFATE, 9-10.5% MONOMETHYLETHER PEG5K AND 0.1M MES IN THE PH RANGE 5.3-6.5 |
-Data collection
| Diffraction | Mean temperature: 87 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 17-ID / Wavelength: 1 Å / Beamline: 17-ID / Wavelength: 1 Å |
| Detector | Type: DECTRIS PILATUS3 6M / Detector: PIXEL / Date: Jun 23, 2011 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 1.83→52.72 Å / Num. obs: 28124 / % possible obs: 99.4 % / Redundancy: 6.5 % / Rmerge(I) obs: 0.057 / Rpim(I) all: 0.026 / Net I/σ(I): 23.3 |
| Reflection shell | Resolution: 1.83→1.93 Å / Redundancy: 6.7 % / Rmerge(I) obs: 0.47 / Num. unique obs: 4020 / Rpim(I) all: 0.211 / % possible all: 98.8 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  FOURIER SYNTHESIS FOURIER SYNTHESISStarting model: 2xp2 Resolution: 1.831→52.72 Å / Cor.coef. Fo:Fc: 0.936 / Cor.coef. Fo:Fc free: 0.922 / SU R Cruickshank DPI: 0.135 / Cross valid method: THROUGHOUT / SU R Blow DPI: 0.146 / SU Rfree Blow DPI: 0.128 / SU Rfree Cruickshank DPI: 0.122
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 27.76 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.23 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.831→52.72 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.831→1.84 Å
|
 Movie
Movie Controller
Controller


 PDBj
PDBj