[English] 日本語
 Yorodumi
Yorodumi- PDB-7ot3: Human Prolyl-tRNA Synthetase in Complex with L-proline and Compound 3b -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7ot3 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human Prolyl-tRNA Synthetase in Complex with L-proline and Compound 3b | ||||||||||||
 Components Components | Bifunctional glutamate/proline--tRNA ligase | ||||||||||||
 Keywords Keywords | LIGASE / beta barrel / tRNA synthetase / protein-inhibitor complex | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of long-chain fatty acid import into cell / Selenoamino acid metabolism / glutamate-tRNA ligase / glutamate-tRNA ligase activity / proline-tRNA ligase / proline-tRNA ligase activity / prolyl-tRNA aminoacylation / glutamyl-tRNA aminoacylation / tRNA modification in the nucleus and cytosol / Cytosolic tRNA aminoacylation ...regulation of long-chain fatty acid import into cell / Selenoamino acid metabolism / glutamate-tRNA ligase / glutamate-tRNA ligase activity / proline-tRNA ligase / proline-tRNA ligase activity / prolyl-tRNA aminoacylation / glutamyl-tRNA aminoacylation / tRNA modification in the nucleus and cytosol / Cytosolic tRNA aminoacylation / aminoacyl-tRNA synthetase multienzyme complex / tRNA aminoacylation for protein translation / GAIT complex / Transcriptional and post-translational regulation of MITF-M expression and activity / RNA stem-loop binding / cellular response to type II interferon / cellular response to insulin stimulus / GTPase binding / sperm midpiece / negative regulation of translation / ribonucleoprotein complex / protein homodimerization activity / zinc ion binding / ATP binding / membrane / identical protein binding / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.53 Å MOLECULAR REPLACEMENT / Resolution: 2.53 Å | ||||||||||||
 Authors Authors | Pang, L. / Zitko, J. / Strelkov, S.V. / Weeks, S.D. | ||||||||||||
| Funding support |  Belgium, 3items Belgium, 3items
| ||||||||||||
 Citation Citation |  Journal: Int J Mol Sci / Year: 2021 Journal: Int J Mol Sci / Year: 2021Title: Towards Novel 3-Aminopyrazinamide-Based Prolyl-tRNA Synthetase Inhibitors: In Silico Modelling, Thermal Shift Assay and Structural Studies. Authors: Pang, L. / Weeks, S.D. / Juhas, M. / Strelkov, S.V. / Zitko, J. / Van Aerschot, A. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7ot3.cif.gz 7ot3.cif.gz | 401.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7ot3.ent.gz pdb7ot3.ent.gz | 328.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7ot3.json.gz 7ot3.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ot/7ot3 https://data.pdbj.org/pub/pdb/validation_reports/ot/7ot3 ftp://data.pdbj.org/pub/pdb/validation_reports/ot/7ot3 ftp://data.pdbj.org/pub/pdb/validation_reports/ot/7ot3 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7osyC  7oszC  7ot0C  7ot1C  7ot2C  5vadS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 2 molecules AB
| #1: Protein | Mass: 58087.375 Da / Num. of mol.: 2 / Fragment: Prolyl-tRNA synthetase Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: EPRS1, EPRS, GLNS, PARS, QARS, QPRS, PIG32 / Plasmid: pETRUK / Production host: Homo sapiens (human) / Gene: EPRS1, EPRS, GLNS, PARS, QARS, QPRS, PIG32 / Plasmid: pETRUK / Production host:  References: UniProt: P07814, glutamate-tRNA ligase, proline-tRNA ligase |
|---|
-Non-polymers , 5 types, 36 molecules 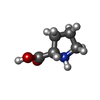
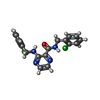







| #2: Chemical | | #3: Chemical | #4: Chemical | #5: Chemical | ChemComp-SR / #6: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.36 Å3/Da / Density % sol: 47.89 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: microbatch / pH: 7.5 Details: The purified protein at 30 mg/mL in 10 mM Tris pH 7, 100 mM NaCl, 2.5 mM 2-mercaptoethanol was incubated with 10 mM L-proline, 2 mM compound and 12% (v/v) DMSO on ice for 1 hr. Crystals were ...Details: The purified protein at 30 mg/mL in 10 mM Tris pH 7, 100 mM NaCl, 2.5 mM 2-mercaptoethanol was incubated with 10 mM L-proline, 2 mM compound and 12% (v/v) DMSO on ice for 1 hr. Crystals were grown in a Terasaki Microbatch plate by mixing 1 uL the pre-mix with 1 uL of reservoir solution containing 0.25-0.4 M SrCl2, 15-20% (v/v) PEG3350 and 100 mM HEPES pH 7.5. The drops were covered with paraffin oil. Crystals were flash frozen in liquid nitrogen directly from the plate. |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06SA / Wavelength: 1.000021 Å / Beamline: X06SA / Wavelength: 1.000021 Å | ||||||||||||||||||||||||||||||
| Detector | Type: DECTRIS EIGER X 16M / Detector: PIXEL / Date: Apr 22, 2021 | ||||||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 1.000021 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.48→54.77 Å / Num. obs: 38157 / % possible obs: 99.2 % / Redundancy: 6.9 % / CC1/2: 0.999 / Rmerge(I) obs: 0.047 / Rpim(I) all: 0.019 / Rrim(I) all: 0.051 / Net I/σ(I): 17.1 / Num. measured all: 264382 | ||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5VAD Resolution: 2.53→54.77 Å / SU ML: 0.44 / Cross valid method: THROUGHOUT / σ(F): 1.38 / Phase error: 32.99 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 357.9 Å2 / Biso mean: 115.1964 Å2 / Biso min: 44.6 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.53→54.77 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0 / Total num. of bins used: 13
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj








