[English] 日本語
 Yorodumi
Yorodumi- PDB-6zog: Minocycline binding to the deep binding pocket of AcrB-I38F_I671T -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6zog | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Minocycline binding to the deep binding pocket of AcrB-I38F_I671T | ||||||||||||
 Components Components |
| ||||||||||||
 Keywords Keywords | TRANSPORT PROTEIN / Multidrug efflux pump / Membrane protein | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationalkane transmembrane transporter activity / alkane transport / enterobactin transport / enterobactin transmembrane transporter activity / xenobiotic detoxification by transmembrane export across the cell outer membrane / periplasmic side of plasma membrane / efflux pump complex / Iron assimilation using enterobactin / bile acid transmembrane transporter activity / Antimicrobial resistance ...alkane transmembrane transporter activity / alkane transport / enterobactin transport / enterobactin transmembrane transporter activity / xenobiotic detoxification by transmembrane export across the cell outer membrane / periplasmic side of plasma membrane / efflux pump complex / Iron assimilation using enterobactin / bile acid transmembrane transporter activity / Antimicrobial resistance / bile acid and bile salt transport / Secretion of toxins / efflux transmembrane transporter activity / xenobiotic transmembrane transporter activity / fatty acid transport / xenobiotic transport / response to toxic substance / response to xenobiotic stimulus / response to antibiotic / membrane / identical protein binding / plasma membrane Similarity search - Function | ||||||||||||
| Biological species |  synthetic construct (others) | ||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 2.75 Å molecular replacement / Resolution: 2.75 Å | ||||||||||||
 Authors Authors | Tam, H.K. / Foong, W.E. / Pos, K.M. | ||||||||||||
| Funding support |  Germany, European Union, 3items Germany, European Union, 3items
| ||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2021 Journal: Nat Commun / Year: 2021Title: Allosteric drug transport mechanism of multidrug transporter AcrB. Authors: Tam, H.K. / Foong, W.E. / Oswald, C. / Herrmann, A. / Zeng, H. / Pos, K.M. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6zog.cif.gz 6zog.cif.gz | 664.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6zog.ent.gz pdb6zog.ent.gz | 531.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6zog.json.gz 6zog.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/zo/6zog https://data.pdbj.org/pub/pdb/validation_reports/zo/6zog ftp://data.pdbj.org/pub/pdb/validation_reports/zo/6zog ftp://data.pdbj.org/pub/pdb/validation_reports/zo/6zog | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6zo5C  6zo6C  6zo7C  6zo8C  6zo9C  6zoaC  6zobC  6zocC  6zodC  6zoeC  6zofC  6zohC  5jmnS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 2 types, 5 molecules ABCDE
| #1: Protein | Mass: 114758.258 Da / Num. of mol.: 3 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #2: Protein | Mass: 18317.566 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.) synthetic construct (others) / Gene: ARTIFICIAL GENE / Plasmid: PQE30 / Production host:  |
|---|
-Sugars , 1 types, 6 molecules 
| #3: Sugar | ChemComp-LMT / |
|---|
-Non-polymers , 14 types, 158 molecules 




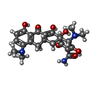

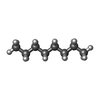




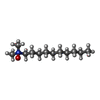














| #4: Chemical | | #5: Chemical | ChemComp-EDO / #6: Chemical | ChemComp-DDR / ( | #7: Chemical | ChemComp-MYS / | #8: Chemical | ChemComp-8K6 / | #9: Chemical | ChemComp-MIY / ( | #10: Chemical | ChemComp-GOL / #11: Chemical | ChemComp-OCT / | #12: Chemical | #13: Chemical | ChemComp-D12 / | #14: Chemical | ChemComp-PTY / | #15: Chemical | ChemComp-C14 / | #16: Chemical | ChemComp-DDQ / | #17: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.67 Å3/Da / Density % sol: 66.53 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, hanging drop / pH: 6.6 Details: 0.05M ADA, PH 6.6, 0.15-0.25M AMMONIUM SULFATE, 5% GLYCEROL, 8-9% PEG4000, 0.002M MINOCYCLINE |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  PETRA III, EMBL c/o DESY PETRA III, EMBL c/o DESY  / Beamline: P13 (MX1) / Wavelength: 1.033 Å / Beamline: P13 (MX1) / Wavelength: 1.033 Å | |||||||||||||||||||||||||||
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Oct 14, 2016 | |||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 1.033 Å / Relative weight: 1 | |||||||||||||||||||||||||||
| Reflection | Resolution: 2.75→49.59 Å / Num. obs: 151127 / % possible obs: 100 % / Redundancy: 7.6 % / CC1/2: 0.99 / Rmerge(I) obs: 0.191 / Rpim(I) all: 0.074 / Rrim(I) all: 0.205 / Net I/σ(I): 9.8 / Num. measured all: 1141557 / Scaling rejects: 256 | |||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
-Phasing
| Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5JMN Resolution: 2.75→49.59 Å / Cor.coef. Fo:Fc: 0.91 / Cor.coef. Fo:Fc free: 0.882 / SU B: 18.141 / SU ML: 0.321 / SU R Cruickshank DPI: 0.5452 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.545 / ESU R Free: 0.323 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS U VALUES : REFINED INDIVIDUALLY
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 151.54 Å2 / Biso mean: 61.673 Å2 / Biso min: 26 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.75→49.59 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.75→2.821 Å / Rfactor Rfree error: 0 / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller












 PDBj
PDBj
















