[English] 日本語
 Yorodumi
Yorodumi- PDB-6ryf: High-resolution crystal structure of ERAP1 in complex with 15mer ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6ryf | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | High-resolution crystal structure of ERAP1 in complex with 15mer phosphinic peptide | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | HYDROLASE / endoplasmic reticulum aminopeptidase 1 / ERAP1 / antigen presentation | |||||||||
| Function / homology |  Function and homology information Function and homology informationinterleukin-1, type II receptor binding / interleukin-6 receptor binding / Hydrolases; Acting on peptide bonds (peptidases); Aminopeptidases / Synthesis and processing of ENV and VPU / symbiont-mediated evasion of host immune response / metalloexopeptidase activity / positive regulation of establishment of T cell polarity / Alpha-defensins / peptide catabolic process / regulation of innate immune response ...interleukin-1, type II receptor binding / interleukin-6 receptor binding / Hydrolases; Acting on peptide bonds (peptidases); Aminopeptidases / Synthesis and processing of ENV and VPU / symbiont-mediated evasion of host immune response / metalloexopeptidase activity / positive regulation of establishment of T cell polarity / Alpha-defensins / peptide catabolic process / regulation of innate immune response / Dectin-2 family / metalloaminopeptidase activity / membrane protein ectodomain proteolysis / Binding and entry of HIV virion / aminopeptidase activity / antigen processing and presentation of peptide antigen via MHC class I / fat cell differentiation / positive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / host cell endosome membrane / actin filament organization / Antigen Presentation: Folding, assembly and peptide loading of class I MHC / response to bacterium / Assembly Of The HIV Virion / antigen processing and presentation of endogenous peptide antigen via MHC class I / Budding and maturation of HIV virion / regulation of blood pressure / positive regulation of angiogenesis / angiogenesis / endopeptidase activity / clathrin-dependent endocytosis of virus by host cell / adaptive immune response / viral protein processing / endoplasmic reticulum lumen / receptor ligand activity / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / symbiont entry into host cell / endoplasmic reticulum membrane / virion attachment to host cell / host cell plasma membrane / virion membrane / structural molecule activity / endoplasmic reticulum / proteolysis / : / extracellular exosome / extracellular region / zinc ion binding / membrane / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human)  Human immunodeficiency virus 1 Human immunodeficiency virus 1 | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.72 Å MOLECULAR REPLACEMENT / Resolution: 1.72 Å | |||||||||
 Authors Authors | Giastas, P. / Stratikos, E. | |||||||||
 Citation Citation |  Journal: Proc.Natl.Acad.Sci.USA / Year: 2019 Journal: Proc.Natl.Acad.Sci.USA / Year: 2019Title: Mechanism for antigenic peptide selection by endoplasmic reticulum aminopeptidase 1. Authors: Giastas, P. / Mpakali, A. / Papakyriakou, A. / Lelis, A. / Kokkala, P. / Neu, M. / Rowland, P. / Liddle, J. / Georgiadis, D. / Stratikos, E. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6ryf.cif.gz 6ryf.cif.gz | 391.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6ryf.ent.gz pdb6ryf.ent.gz | 317.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6ryf.json.gz 6ryf.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ry/6ryf https://data.pdbj.org/pub/pdb/validation_reports/ry/6ryf ftp://data.pdbj.org/pub/pdb/validation_reports/ry/6ryf ftp://data.pdbj.org/pub/pdb/validation_reports/ry/6ryf | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6rqxC  2yd0S C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein / Protein/peptide , 2 types, 2 molecules AG
| #1: Protein | Mass: 102208.523 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: ERAP1, APPILS, ARTS1, KIAA0525, UNQ584/PRO1154 Homo sapiens (human) / Gene: ERAP1, APPILS, ARTS1, KIAA0525, UNQ584/PRO1154Production host:  Spodoptera aff. frugiperda 1 BOLD-2017 (butterflies/moths) Spodoptera aff. frugiperda 1 BOLD-2017 (butterflies/moths)References: UniProt: Q9NZ08, Hydrolases; Acting on peptide bonds (peptidases); Aminopeptidases |
|---|---|
| #2: Protein/peptide | Mass: 1767.086 Da / Num. of mol.: 1 / Source method: obtained synthetically / Details: SYNTHETIC PHOSPHINIC PSEUDOPEPTIDE / Source: (synth.)   Human immunodeficiency virus 1 / References: UniProt: P04578*PLUS Human immunodeficiency virus 1 / References: UniProt: P04578*PLUS |
-Sugars , 2 types, 4 molecules 
| #3: Polysaccharide | Source method: isolated from a genetically manipulated source #9: Sugar | |
|---|
-Non-polymers , 6 types, 617 molecules 



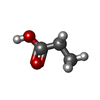






| #4: Chemical | ChemComp-ZN / | ||||||
|---|---|---|---|---|---|---|---|
| #5: Chemical | ChemComp-B3P / | ||||||
| #6: Chemical | ChemComp-EDO / #7: Chemical | ChemComp-NA / #8: Chemical | ChemComp-PPI / | #10: Water | ChemComp-HOH / | |
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.44 Å3/Da / Density % sol: 49.69 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop Details: 100 mM propionic acid, cacodylate and Bis-tris propane pH 7.0, 25% PEG 1500 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  PETRA III, EMBL c/o DESY PETRA III, EMBL c/o DESY  / Beamline: P13 (MX1) / Wavelength: 0.9763 Å / Beamline: P13 (MX1) / Wavelength: 0.9763 Å |
| Detector | Type: DECTRIS PILATUS 2M / Detector: PIXEL / Date: Jun 24, 2018 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9763 Å / Relative weight: 1 |
| Reflection | Resolution: 1.72→91.52 Å / Num. obs: 106245 / % possible obs: 99.98 % / Redundancy: 8 % / CC1/2: 0.999 / Net I/σ(I): 12.5 |
| Reflection shell | Resolution: 1.72→1.75 Å / Num. unique obs: 5264 / CC1/2: 0.368 / % possible all: 99.98 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 2YD0 Resolution: 1.72→91.52 Å / SU ML: 0.21 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 21.42
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.72→91.52 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj















