+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6kmu | ||||||
|---|---|---|---|---|---|---|---|
| Title | P22/P10 complex of caspase-11 mutant C254A | ||||||
 Components Components | (Caspase-4) x 5 | ||||||
 Keywords Keywords | IMMUNE SYSTEM / pyroptosis | ||||||
| Function / homology |  Function and homology information Function and homology informationcaspase-11 / non-canonical inflammasome complex / Pyroptosis / positive regulation of interleukin-18-mediated signaling pathway / pyroptotic cell death / non-canonical inflammasome complex assembly / detection of lipopolysaccharide / canonical inflammasome complex / NOD1/2 Signaling Pathway / positive regulation of macrophage cytokine production ...caspase-11 / non-canonical inflammasome complex / Pyroptosis / positive regulation of interleukin-18-mediated signaling pathway / pyroptotic cell death / non-canonical inflammasome complex assembly / detection of lipopolysaccharide / canonical inflammasome complex / NOD1/2 Signaling Pathway / positive regulation of macrophage cytokine production / pyroptotic inflammatory response / positive regulation of NLRP3 inflammasome complex assembly / protein autoprocessing / ectopic germ cell programmed cell death / actin filament organization / protein maturation / positive regulation of interleukin-1 beta production / lipopolysaccharide binding / positive regulation of inflammatory response / regulation of inflammatory response / regulation of apoptotic process / defense response to Gram-positive bacterium / defense response to bacterium / innate immune response / cysteine-type endopeptidase activity / lipid binding / endoplasmic reticulum membrane / mitochondrion / extracellular region / cytosol / cytoplasm Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.1 Å MOLECULAR REPLACEMENT / Resolution: 2.1 Å | ||||||
 Authors Authors | Ding, J. / Sun, Q. | ||||||
 Citation Citation |  Journal: Cell / Year: 2020 Journal: Cell / Year: 2020Title: Structural Mechanism for GSDMD Targeting by Autoprocessed Caspases in Pyroptosis. Authors: Wang, K. / Sun, Q. / Zhong, X. / Zeng, M. / Zeng, H. / Shi, X. / Li, Z. / Wang, Y. / Zhao, Q. / Shao, F. / Ding, J. | ||||||
| History |
|
- Structure visualization
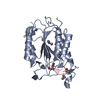
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6kmu.cif.gz 6kmu.cif.gz | 217.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6kmu.ent.gz pdb6kmu.ent.gz | 172 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6kmu.json.gz 6kmu.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  6kmu_validation.pdf.gz 6kmu_validation.pdf.gz | 486.6 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  6kmu_full_validation.pdf.gz 6kmu_full_validation.pdf.gz | 495.8 KB | Display | |
| Data in XML |  6kmu_validation.xml.gz 6kmu_validation.xml.gz | 38.9 KB | Display | |
| Data in CIF |  6kmu_validation.cif.gz 6kmu_validation.cif.gz | 54.4 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/km/6kmu https://data.pdbj.org/pub/pdb/validation_reports/km/6kmu ftp://data.pdbj.org/pub/pdb/validation_reports/km/6kmu ftp://data.pdbj.org/pub/pdb/validation_reports/km/6kmu | HTTPS FTP |
-Related structure data
| Related structure data |  6kmtSC  6kmvC  6kmzC  6kn0C  6kn1C S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| 3 | 
| ||||||||
| 4 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 5 types, 8 molecules AEBFHCDG
| #1: Protein | Mass: 20783.809 Da / Num. of mol.: 2 / Mutation: C254A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #2: Protein | Mass: 10113.619 Da / Num. of mol.: 3 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #3: Protein | | Mass: 20884.914 Da / Num. of mol.: 1 / Mutation: C254A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #4: Protein | | Mass: 10212.750 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #5: Protein | | Mass: 17408.006 Da / Num. of mol.: 1 / Mutation: C254A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Non-polymers , 1 types, 364 molecules 
| #6: Water | ChemComp-HOH / |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.74 Å3/Da / Density % sol: 55.12 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop Details: 100 mM HEPES (pH 7.5), 12% (w/v) polyethylene glycol 3350, and 4% formaldehyde |
-Data collection
| Diffraction | Mean temperature: 93 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: NFPSS SYNCHROTRON / Site: NFPSS  / Beamline: BL19U1 / Wavelength: 0.97892 Å / Beamline: BL19U1 / Wavelength: 0.97892 Å |
| Detector | Type: DECTRIS PILATUS3 6M / Detector: PIXEL / Date: Oct 2, 2018 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.97892 Å / Relative weight: 1 |
| Reflection | Resolution: 2.1→49.78 Å / Num. obs: 71527 / % possible obs: 97.7 % / Redundancy: 3.41 % / CC1/2: 0.997 / Rmerge(I) obs: 0.057 / Net I/σ(I): 11.81 |
| Reflection shell | Resolution: 2.1→2.15 Å / Rmerge(I) obs: 0.369 / Mean I/σ(I) obs: 2.72 / Num. unique obs: 5274 / CC1/2: 0.915 |
- Processing
Processing
| Software |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 6kmt Resolution: 2.1→49.78 Å / Cross valid method: FREE R-VALUE
| ||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.1→49.78 Å
| ||||||||||||||||
| LS refinement shell | Resolution: 2.0994→2.1519 Å
|
 Movie
Movie Controller
Controller












 PDBj
PDBj




