+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6hv5 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Yeast 20S proteasome with human beta2i (1-53) in complex with 4 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | HYDROLASE / Proteasome / Mutant / Inhibitor / Binding Analysis | ||||||
| Function / homology |  Function and homology information Function and homology informationspermatoproteasome complex / antigen processing and presentation of exogenous peptide antigen via MHC class I, TAP-dependent / Antigen processing: Ub, ATP-independent proteasomal degradation / ER-Phagosome pathway / Antigen processing: Ub, ATP-independent proteasomal degradation / proteasome core complex assembly / Proteasome assembly / Cross-presentation of soluble exogenous antigens (endosomes) / TNFR2 non-canonical NF-kB pathway / nuclear outer membrane-endoplasmic reticulum membrane network ...spermatoproteasome complex / antigen processing and presentation of exogenous peptide antigen via MHC class I, TAP-dependent / Antigen processing: Ub, ATP-independent proteasomal degradation / ER-Phagosome pathway / Antigen processing: Ub, ATP-independent proteasomal degradation / proteasome core complex assembly / Proteasome assembly / Cross-presentation of soluble exogenous antigens (endosomes) / TNFR2 non-canonical NF-kB pathway / nuclear outer membrane-endoplasmic reticulum membrane network / Regulation of PTEN stability and activity / Proteasome assembly / CDK-mediated phosphorylation and removal of Cdc6 / proteasome core complex / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / KEAP1-NFE2L2 pathway / Neddylation / Cross-presentation of soluble exogenous antigens (endosomes) / Ubiquitin-Mediated Degradation of Phosphorylated Cdc25A / Orc1 removal from chromatin / MAPK6/MAPK4 signaling / humoral immune response / Antigen processing: Ubiquitination & Proteasome degradation / proteasome storage granule / proteasomal ubiquitin-independent protein catabolic process / T cell proliferation / Ub-specific processing proteases / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / endopeptidase activator activity / threonine-type endopeptidase activity / proteasome assembly / proteasome core complex, alpha-subunit complex / Neutrophil degranulation / proteasome complex / cell morphogenesis / peroxisome / ER-Phagosome pathway / endopeptidase activity / proteasome-mediated ubiquitin-dependent protein catabolic process / mRNA binding / endoplasmic reticulum membrane / mitochondrion / nucleus / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3 Å MOLECULAR REPLACEMENT / Resolution: 3 Å | ||||||
 Authors Authors | Huber, E.M. / Groll, M. | ||||||
| Funding support |  Germany, 1items Germany, 1items
| ||||||
 Citation Citation |  Journal: J.Med.Chem. / Year: 2019 Journal: J.Med.Chem. / Year: 2019Title: Structure-Based Design of Inhibitors Selective for Human Proteasome beta 2c or beta 2i Subunits. Authors: Xin, B.T. / Huber, E.M. / de Bruin, G. / Heinemeyer, W. / Maurits, E. / Espinal, C. / Du, Y. / Janssens, M. / Weyburne, E.S. / Kisselev, A.F. / Florea, B.I. / Driessen, C. / van der Marel, G. ...Authors: Xin, B.T. / Huber, E.M. / de Bruin, G. / Heinemeyer, W. / Maurits, E. / Espinal, C. / Du, Y. / Janssens, M. / Weyburne, E.S. / Kisselev, A.F. / Florea, B.I. / Driessen, C. / van der Marel, G.A. / Groll, M. / Overkleeft, H.S. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6hv5.cif.gz 6hv5.cif.gz | 2.4 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6hv5.ent.gz pdb6hv5.ent.gz | 2 MB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6hv5.json.gz 6hv5.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/hv/6hv5 https://data.pdbj.org/pub/pdb/validation_reports/hv/6hv5 ftp://data.pdbj.org/pub/pdb/validation_reports/hv/6hv5 ftp://data.pdbj.org/pub/pdb/validation_reports/hv/6hv5 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6htbC  6htcC  6htdC  6htpC  6htrC  6hubC  6hucC  6huqC  6huuC  6huvC  6hv3C  6hv4C  6hv7C  6hvaC  6hvrC  6hvsC  6hvtC  6hvuC  6hvvC  6hvwC  6hvxC  6hvyC  6hw0C  6hw3C  6hw4C  6hw5C  6hw6C  6hw7C  6hw8C  6hw9C  6hwaC  6hwbC  6hwcC  6hwdC  6hweC  6hwfC  5cz4S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unit cell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments:
NCS ensembles :
NCS oper:
|
- Components
Components
-Proteasome subunit alpha type- ... , 6 types, 12 molecules AOBPCQDRESGU
| #1: Protein | Mass: 27191.828 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P23639, proteasome endopeptidase complex #2: Protein | Mass: 28748.230 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P23638, proteasome endopeptidase complex #3: Protein | Mass: 28478.111 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P40303, proteasome endopeptidase complex #4: Protein | Mass: 28649.086 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P32379, proteasome endopeptidase complex #5: Protein | Mass: 25634.000 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P40302, proteasome endopeptidase complex #7: Protein | Mass: 28033.830 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P21243, proteasome endopeptidase complex |
|---|
-Protein , 1 types, 2 molecules FT
| #6: Protein | Mass: 31575.068 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P21242, proteasome endopeptidase complex |
|---|
-Proteasome subunit beta type- ... , 7 types, 14 molecules HVIWJXKYLZMaNb
| #8: Protein | Mass: 24443.674 Da / Num. of mol.: 2 Mutation: Chimera: 1-53 Homo sapiens,Chimera: 1-53 Homo sapiens Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human), (gene. exp.) Homo sapiens (human), (gene. exp.)  Gene: PSMB10, LMP10, MECL1, PUP1, YOR157C / Production host:  References: UniProt: P40306, UniProt: P25043, proteasome endopeptidase complex #9: Protein | Mass: 22627.842 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P25451, proteasome endopeptidase complex #10: Protein | Mass: 22545.676 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P22141, proteasome endopeptidase complex #11: Protein | Mass: 23325.248 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P30656, proteasome endopeptidase complex #12: Protein | Mass: 24883.928 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P23724, proteasome endopeptidase complex #13: Protein | Mass: 27200.893 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P30657, proteasome endopeptidase complex #14: Protein | Mass: 21517.186 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P38624, proteasome endopeptidase complex |
|---|
-Non-polymers , 4 types, 262 molecules 

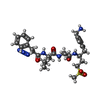




| #15: Chemical | ChemComp-MG / #16: Chemical | #17: Chemical | ChemComp-GQH / ( #18: Water | ChemComp-HOH / | |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.67 Å3/Da / Density % sol: 66.52 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 6.8 / Details: 20 mM MgAC2, 13% MPD, 0.1 M MES / PH range: 6.8 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06SA / Wavelength: 1 Å / Beamline: X06SA / Wavelength: 1 Å |
| Detector | Type: DECTRIS EIGER X 16M / Detector: PIXEL / Date: Nov 20, 2016 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 3→50 Å / Num. obs: 205492 / % possible obs: 97.6 % / Redundancy: 3.2 % / Rmerge(I) obs: 0.081 / Net I/σ(I): 12.5 |
| Reflection shell | Resolution: 3→3.1 Å / Rmerge(I) obs: 0.57 / Mean I/σ(I) obs: 3.2 / % possible all: 99.3 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5CZ4 Resolution: 3→15 Å / Cor.coef. Fo:Fc: 0.961 / Cor.coef. Fo:Fc free: 0.943 / SU B: 40.341 / SU ML: 0.293 / Cross valid method: THROUGHOUT / ESU R Free: 0.319 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 78.206 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3→15 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller























 PDBj
PDBj
















