[English] 日本語
 Yorodumi
Yorodumi- PDB-6h60: pseudo-atomic structural model of the E3BP component of the human... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6h60 | ||||||
|---|---|---|---|---|---|---|---|
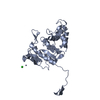
| Title | pseudo-atomic structural model of the E3BP component of the human pyruvate dehydrogenase multienzyme complex | ||||||
 Components Components | Pyruvate dehydrogenase protein X component, mitochondrial | ||||||
 Keywords Keywords | OXIDOREDUCTASE / Pyruvate dehydrogenase / human | ||||||
| Function / homology |  Function and homology information Function and homology informationPDH complex synthesizes acetyl-CoA from PYR / dihydrolipoyllysine-residue acetyltransferase activity / Regulation of pyruvate dehydrogenase (PDH) complex / pyruvate decarboxylation to acetyl-CoA / pyruvate dehydrogenase complex / Signaling by Retinoic Acid / mitochondrial matrix / mitochondrion / nucleoplasm / plasma membrane Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 6 Å | ||||||
 Authors Authors | Haselbach, D. / Prajapati, S. / Tittmann, K. / Stark, H. | ||||||
| Funding support |  Germany, 1items Germany, 1items
| ||||||
 Citation Citation |  Journal: Structure / Year: 2019 Journal: Structure / Year: 2019Title: Structural and Functional Analyses of the Human PDH Complex Suggest a "Division-of-Labor" Mechanism by Local E1 and E3 Clusters. Authors: Sabin Prajapati / David Haselbach / Sabine Wittig / Mulchand S Patel / Ashwin Chari / Carla Schmidt / Holger Stark / Kai Tittmann /   Abstract: The pseudo-atomic structural model of human pyruvate dehydrogenase complex (PDHc) core composed of full-length E2 and E3BP components, calculated from our cryoelectron microscopy-derived density maps ...The pseudo-atomic structural model of human pyruvate dehydrogenase complex (PDHc) core composed of full-length E2 and E3BP components, calculated from our cryoelectron microscopy-derived density maps at 6-Å resolution, is similar to those of prokaryotic E2 structures. The spatial organization of human PDHc components as evidenced by negative-staining electron microscopy and native mass spectrometry is not homogeneous, and entails the unanticipated formation of local clusters of E1:E2 and E3BP:E3 complexes. Such uneven, clustered organization translates into specific duties for E1-E2 clusters (oxidative decarboxylation and acetyl transfer) and E3BP-E3 clusters (regeneration of reduced lipoamide) corresponding to half-reactions of the PDHc catalytic cycle. The addition of substrate coenzyme A modulates the conformational landscape of PDHc, in particular of the lipoyl domains, extending the postulated multiple random coupling mechanism. The conformational and associated chemical landscapes of PDHc are thus not determined entirely stochastically, but are restrained and channeled through an asymmetric architecture and further modulated by substrate binding. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6h60.cif.gz 6h60.cif.gz | 47.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6h60.ent.gz pdb6h60.ent.gz | 22.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6h60.json.gz 6h60.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/h6/6h60 https://data.pdbj.org/pub/pdb/validation_reports/h6/6h60 ftp://data.pdbj.org/pub/pdb/validation_reports/h6/6h60 ftp://data.pdbj.org/pub/pdb/validation_reports/h6/6h60 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  0138MC  6h55C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 54191.145 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: PDHX, PDX1 / Production host: Homo sapiens (human) / Gene: PDHX, PDX1 / Production host:  |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: 1 / Type: COMPLEX / Entity ID: all / Source: RECOMBINANT |
|---|---|
| Molecular weight | Experimental value: NO |
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  |
| Buffer solution | pH: 8 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN |
| Electron lens | Mode: BRIGHT FIELD |
| Image recording | Electron dose: 41 e/Å2 / Film or detector model: FEI FALCON II (4k x 4k) |
- Processing
Processing
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
|---|---|
| Symmetry | Point symmetry: I (icosahedral) |
| 3D reconstruction | Resolution: 6 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 29788 / Symmetry type: POINT |
 Movie
Movie Controller
Controller









 PDBj
PDBj


