Entry Database : PDB / ID : 6bdjTitle Crystal structure of dioxygenase Tetur07g02040 Tetur07g02040 Keywords / / / / Function / homology / / / / / / Biological species Tetranychus urticae (two-spotted spider mite)Method / / / Resolution : 2.15 Å Authors Daneshian, L. / Schlachter, C.R. / Chruszcz, M. Journal : Insect Biochem.Mol.Biol. / Year : 2019Title : Structural and functional characterization of an intradiol ring-cleavage dioxygenase from the polyphagous spider mite herbivore Tetranychus urticae Koch.Authors : Schlachter, C.R. / Daneshian, L. / Amaya, J. / Klapper, V. / Wybouw, N. / Borowski, T. / Van Leeuwen, T. / Grbic, V. / Grbic, M. / Makris, T.M. / Chruszcz, M. History Deposition Oct 23, 2017 Deposition site / Processing site Revision 1.0 Nov 14, 2018 Provider / Type Revision 1.1 May 29, 2019 Group / Database references / Category / citation_authorItem _citation.country / _citation.journal_abbrev ... _citation.country / _citation.journal_abbrev / _citation.journal_id_CSD / _citation.journal_id_ISSN / _citation.journal_volume / _citation.page_first / _citation.page_last / _citation.pdbx_database_id_DOI / _citation.pdbx_database_id_PubMed / _citation.title / _citation.year Revision 1.2 Nov 6, 2024 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Refinement description / Structure summary Category chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_entry_details / pdbx_modification_feature / struct_conn / struct_ncs_dom_lim Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_ptnr1_label_alt_id / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_ncs_dom_lim.beg_auth_comp_id / _struct_ncs_dom_lim.beg_label_asym_id / _struct_ncs_dom_lim.beg_label_comp_id / _struct_ncs_dom_lim.beg_label_seq_id / _struct_ncs_dom_lim.end_auth_comp_id / _struct_ncs_dom_lim.end_label_asym_id / _struct_ncs_dom_lim.end_label_comp_id / _struct_ncs_dom_lim.end_label_seq_id
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Tetranychus urticae (two-spotted spider mite)
Tetranychus urticae (two-spotted spider mite) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.15 Å
MOLECULAR REPLACEMENT / Resolution: 2.15 Å  Authors
Authors Citation
Citation Journal: Insect Biochem.Mol.Biol. / Year: 2019
Journal: Insect Biochem.Mol.Biol. / Year: 2019 Structure visualization
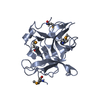
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 6bdj.cif.gz
6bdj.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb6bdj.ent.gz
pdb6bdj.ent.gz PDB format
PDB format 6bdj.json.gz
6bdj.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/bd/6bdj
https://data.pdbj.org/pub/pdb/validation_reports/bd/6bdj ftp://data.pdbj.org/pub/pdb/validation_reports/bd/6bdj
ftp://data.pdbj.org/pub/pdb/validation_reports/bd/6bdj Links
Links Assembly
Assembly


 Components
Components Tetranychus urticae (two-spotted spider mite)
Tetranychus urticae (two-spotted spider mite)
 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  APS
APS  / Beamline: 22-ID / Wavelength: 1 Å
/ Beamline: 22-ID / Wavelength: 1 Å Processing
Processing MOLECULAR REPLACEMENT / Resolution: 2.15→40 Å / Cor.coef. Fo:Fc: 0.955 / Cor.coef. Fo:Fc free: 0.941 / Cross valid method: THROUGHOUT / ESU R: 0.274 / ESU R Free: 0.208 / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
MOLECULAR REPLACEMENT / Resolution: 2.15→40 Å / Cor.coef. Fo:Fc: 0.955 / Cor.coef. Fo:Fc free: 0.941 / Cross valid method: THROUGHOUT / ESU R: 0.274 / ESU R Free: 0.208 / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS Movie
Movie Controller
Controller













 PDBj
PDBj

