+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5tss | ||||||
|---|---|---|---|---|---|---|---|
| Title | TOXIC SHOCK SYNDROME TOXIN-1: ORTHORHOMBIC P222(1) CRYSTAL FORM | ||||||
 Components Components | TOXIC SHOCK SYNDROME TOXIN-1 | ||||||
 Keywords Keywords | TOXIN / SUPERANTIGEN | ||||||
| Function / homology |  Function and homology information Function and homology information | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.9 Å MOLECULAR REPLACEMENT / Resolution: 2.9 Å | ||||||
 Authors Authors | Prasad, G.S. / Radhakrishnan, R. / Mitchell, D.T. / Earhart, C.A. / Dinges, M.M. / Cook, W.J. / Schlivert, P.M. / Ohlendorf, D.H. | ||||||
 Citation Citation |  Journal: Protein Sci. / Year: 1997 Journal: Protein Sci. / Year: 1997Title: Refined structures of three crystal forms of toxic shock syndrome toxin-1 and of a tetramutant with reduced activity. Authors: Prasad, G.S. / Radhakrishnan, R. / Mitchell, D.T. / Earhart, C.A. / Dinges, M.M. / Cook, W.J. / Schlievert, P.M. / Ohlendorf, D.H. #1:  Journal: Biochemistry / Year: 1993 Journal: Biochemistry / Year: 1993Title: Structure of Toxic Shock Syndrome Toxin 1 Authors: Prasad, G.S. / Earhart, C.A. / Murray, D.L. / Novick, R.P. / Schlievert, P.M. / Ohlendorf, D.H. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5tss.cif.gz 5tss.cif.gz | 87 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5tss.ent.gz pdb5tss.ent.gz | 67.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5tss.json.gz 5tss.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ts/5tss https://data.pdbj.org/pub/pdb/validation_reports/ts/5tss ftp://data.pdbj.org/pub/pdb/validation_reports/ts/5tss ftp://data.pdbj.org/pub/pdb/validation_reports/ts/5tss | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 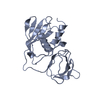
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
| ||||||||
| Noncrystallographic symmetry (NCS) | NCS oper: (Code: given Matrix: (-0.99889, -0.02139, 0.04192), Vector: |
- Components
Components
| #1: Protein | Mass: 22103.770 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #2: Water | ChemComp-HOH / | Sequence details | TSST-1 IS PRODUCED AS A PRO-PEPTIDE WITH 40 AMINO ACIDS AT ITS AMINO-TERMINUS. THE NUMBERING SCHEME ...TSST-1 IS PRODUCED AS A PRO-PEPTIDE WITH 40 AMINO ACIDS AT ITS AMINO-TERMINUS. THE NUMBERING SCHEME HERE BEGINS WITH THE FIRST RESIDUE OF THE MATURE PROTEIN. | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 3 X-RAY DIFFRACTION / Number of used crystals: 3 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.41 Å3/Da / Density % sol: 49 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 6.5 Details: 50 MM TRIS-MALATE BUFFER, PH 5.8-6.6, 24% PEG 4000, 0.5-1 M LICL, pH 6.5 PH range: 5.8-6.6 | ||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 18 ℃ / Method: vapor diffusion / PH range low: 6.6 / PH range high: 5.8 | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 293 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RUH2R / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU RUH2R / Wavelength: 1.5418 |
| Detector | Type: SIEMENS / Detector: AREA DETECTOR / Date: Jul 1, 1992 |
| Radiation | Monochromator: YES / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.9→20 Å / Num. obs: 185649 / % possible obs: 98 % / Redundancy: 18.6 % / Rsym value: 0.108 / Net I/σ(I): 6.6 |
| Reflection shell | Resolution: 2.9→2.98 Å / Redundancy: 10.3 % / Mean I/σ(I) obs: 1 / Rsym value: 0.376 / % possible all: 87 |
| Reflection | *PLUS Rmerge(I) obs: 0.108 |
| Reflection shell | *PLUS % possible obs: 87 % / Rmerge(I) obs: 0.376 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT / Resolution: 2.9→5 Å / Data cutoff high absF: 100000 / Data cutoff low absF: 0.1 / Isotropic thermal model: RESTRAINED / σ(F): 1 MOLECULAR REPLACEMENT / Resolution: 2.9→5 Å / Data cutoff high absF: 100000 / Data cutoff low absF: 0.1 / Isotropic thermal model: RESTRAINED / σ(F): 1 Details: THE OCCUPANCY FOR SER A 11 THROUGH PRO A 117 IS SET TO ZERO BECAUSE OF THE ABSENCE OF 2FO-FC DENSITY. THIS SEGMENT PARTICIPATES IN A CLOSE CONTACT WITH A TWO-FOLD RELATED MOLECULE. THE ...Details: THE OCCUPANCY FOR SER A 11 THROUGH PRO A 117 IS SET TO ZERO BECAUSE OF THE ABSENCE OF 2FO-FC DENSITY. THIS SEGMENT PARTICIPATES IN A CLOSE CONTACT WITH A TWO-FOLD RELATED MOLECULE. THE CONFORMATION PRESENTED IS SIMILAR TO THAT SEEN IN OTHER CRYSTAL FORMS OF TSST-1.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 20 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.25 Å / Luzzati d res low obs: 5 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.9→5 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints NCS | NCS model details: UNRESTRAINED | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.9→2.98 Å / Total num. of bins used: 20
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.1 / Classification: refinement X-PLOR / Version: 3.1 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | *PLUS Rfactor obs: 0.204 |
 Movie
Movie Controller
Controller
















 PDBj
PDBj

