[English] 日本語
 Yorodumi
Yorodumi- PDB-5mzz: Crystal structure of the decarboxylase AibA/AibB in complex with ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5mzz | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the decarboxylase AibA/AibB in complex with 3-methylglutaconate | ||||||
 Components Components | (Glutaconate CoA-transferase family, subunit ...) x 2 | ||||||
 Keywords Keywords | LYASE / decarboxylase / CoA transferase like fold / 3-methylglutaconate | ||||||
| Function / homology | CoA-transferase activity / Coenzyme A transferase family I / Coenzyme A transferase / Coenzyme A transferase / NagB/RpiA transferase-like / 3-methylpent-2-enedioic acid / ACETATE ION / Glutaconate CoA-transferase family, subunit B / Glutaconate CoA-transferase family, subunit A Function and homology information Function and homology information | ||||||
| Biological species |  Myxococcus xanthus (bacteria) Myxococcus xanthus (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  FOURIER SYNTHESIS / Resolution: 2.3 Å FOURIER SYNTHESIS / Resolution: 2.3 Å | ||||||
 Authors Authors | Bock, T. / Luxenburger, E. / Hoffmann, J. / Schuetza, V. / Feiler, C. / Mueller, R. / Blankenfeldt, W. | ||||||
 Citation Citation |  Journal: Angew. Chem. Int. Ed. Engl. / Year: 2017 Journal: Angew. Chem. Int. Ed. Engl. / Year: 2017Title: AibA/AibB Induces an Intramolecular Decarboxylation in Isovalerate Biosynthesis by Myxococcus xanthus. Authors: Bock, T. / Luxenburger, E. / Hoffmann, J. / Schutza, V. / Feiler, C. / Muller, R. / Blankenfeldt, W. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5mzz.cif.gz 5mzz.cif.gz | 359.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5mzz.ent.gz pdb5mzz.ent.gz | 294 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5mzz.json.gz 5mzz.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mz/5mzz https://data.pdbj.org/pub/pdb/validation_reports/mz/5mzz ftp://data.pdbj.org/pub/pdb/validation_reports/mz/5mzz ftp://data.pdbj.org/pub/pdb/validation_reports/mz/5mzz | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5mzwSC  5mzxC  5mzyC  5n00C  5n01C  5n02C  5n03C S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Glutaconate CoA-transferase family, subunit ... , 2 types, 4 molecules ACDB
| #1: Protein | Mass: 28367.838 Da / Num. of mol.: 2 / Mutation: K191A Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Myxococcus xanthus (strain DK 1622) (bacteria) Myxococcus xanthus (strain DK 1622) (bacteria)Strain: DK 1622 / Gene: MXAN_4264 / Production host:  #2: Protein | Mass: 26280.871 Da / Num. of mol.: 2 / Mutation: E200A, E201A Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Myxococcus xanthus (strain DK 1622) (bacteria) Myxococcus xanthus (strain DK 1622) (bacteria)Strain: DK 1622 / Gene: MXAN_4265 / Production host:  |
|---|
-Non-polymers , 4 types, 375 molecules 

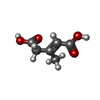




| #3: Chemical | | #4: Chemical | #5: Chemical | #6: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.51 Å3/Da / Density % sol: 50.98 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop Details: 0.085 M sodium acetate, 0.17 M ammonium acetate, 15 % glycerol, 25 % PEG4000 |
-Data collection
| Diffraction | Mean temperature: 100 K | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54 Å ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54 Å | ||||||||||||||||||
| Detector | Type: RIGAKU SATURN 944 / Detector: CCD / Date: Mar 21, 2014 | ||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||
| Radiation wavelength | Wavelength: 1.54 Å / Relative weight: 1 | ||||||||||||||||||
| Reflection | Resolution: 2.3→26.82 Å / Num. obs: 47619 / % possible obs: 99.1 % / Redundancy: 4.2 % / Biso Wilson estimate: 16.59 Å2 / CC1/2: 0.981 / Rmerge(I) obs: 0.178 / Rpim(I) all: 0.099 / Rrim(I) all: 0.204 / Net I/σ(I): 6.6 / Num. measured all: 200861 / Scaling rejects: 0 | ||||||||||||||||||
| Reflection shell |
|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  FOURIER SYNTHESIS FOURIER SYNTHESISStarting model: 5MZW Resolution: 2.3→26.82 Å / SU ML: 0.22 / Cross valid method: FREE R-VALUE / σ(F): 1.35 / Phase error: 19.91
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 63.74 Å2 / Biso mean: 20.3883 Å2 / Biso min: 9.39 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.3→26.82 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0 / Total num. of bins used: 17
|
 Movie
Movie Controller
Controller












 PDBj
PDBj


