[English] 日本語
 Yorodumi
Yorodumi- PDB-5ja2: EntF, a Terminal Nonribosomal Peptide Synthetase Module Bound to ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5ja2 | ||||||
|---|---|---|---|---|---|---|---|
| Title | EntF, a Terminal Nonribosomal Peptide Synthetase Module Bound to the non-Native MbtH-Like Protein PA2412 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | LIGASE / NONRIBOSOMAL PEPTIDE SYNTHETASE / NRPS / CONDENSATION / ADENYLATION / PCP / THIOESTERASE / MBTH-LIKE PROTEIN / PHOSPHOPANTETHEINE / BIOSYNTHETIC PROTEIN | ||||||
| Function / homology |  Function and homology information Function and homology informationL-serine-[L-seryl-carrier protein] ligase / enterobactin synthase / 2,3-dihydroxybenzoate-serine ligase activity / enterobactin synthetase complex / pyoverdine biosynthetic process / enterobactin biosynthetic process / siderophore biosynthetic process / amino acid activation for nonribosomal peptide biosynthetic process / phosphopantetheine binding / nucleotidyltransferase activity ...L-serine-[L-seryl-carrier protein] ligase / enterobactin synthase / 2,3-dihydroxybenzoate-serine ligase activity / enterobactin synthetase complex / pyoverdine biosynthetic process / enterobactin biosynthetic process / siderophore biosynthetic process / amino acid activation for nonribosomal peptide biosynthetic process / phosphopantetheine binding / nucleotidyltransferase activity / ATP binding / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |   | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3 Å MOLECULAR REPLACEMENT / Resolution: 3 Å | ||||||
 Authors Authors | Miller, B.R. / Gulick, A.M. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2016 Journal: J.Biol.Chem. / Year: 2016Title: Structures of a Nonribosomal Peptide Synthetase Module Bound to MbtH-like Proteins Support a Highly Dynamic Domain Architecture. Authors: Miller, B.R. / Drake, E.J. / Shi, C. / Aldrich, C.C. / Gulick, A.M. #1:  Journal: Nature / Year: 2016 Journal: Nature / Year: 2016Title: Structures of two distinct conformations of holo-non-ribosomal peptide synthetases. Authors: Drake, E.J. / Miller, B.R. / Shi, C. / Tarrasch, J.T. / Sundlov, J.A. / Allen, C.L. / Skiniotis, G. / Aldrich, C.C. / Gulick, A.M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5ja2.cif.gz 5ja2.cif.gz | 517.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5ja2.ent.gz pdb5ja2.ent.gz | 421.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5ja2.json.gz 5ja2.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ja/5ja2 https://data.pdbj.org/pub/pdb/validation_reports/ja/5ja2 ftp://data.pdbj.org/pub/pdb/validation_reports/ja/5ja2 ftp://data.pdbj.org/pub/pdb/validation_reports/ja/5ja2 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5ja1SC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 142313.266 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Strain: K12 / Gene: entF, b0586, JW0578 / Variant: W3110 / Plasmid: pET15b / Production host:  References: UniProt: P11454, Transferases; Transferring phosphorus-containing groups; Nucleotidyltransferases | ||||
|---|---|---|---|---|---|
| #2: Protein | Mass: 8649.739 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Pseudomonas aeruginosa (strain ATCC 15692 / PAO1 / 1C / PRS 101 / LMG 12228) (bacteria) Pseudomonas aeruginosa (strain ATCC 15692 / PAO1 / 1C / PRS 101 / LMG 12228) (bacteria)Strain: ATCC 15692 / PAO1 / 1C / PRS 101 / LMG 12228 / Gene: PA2412 / Plasmid: pET15b / Production host:  | ||||
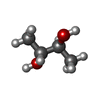
| #3: Chemical | ChemComp-75C / | ||||
| #4: Chemical | | #5: Water | ChemComp-HOH / | Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.98 Å3/Da / Density % sol: 57.2 % |
|---|---|
| Crystal grow | Temperature: 287.15 K / Method: vapor diffusion, hanging drop / pH: 7.5 Details: 100 mM Bis Tris propane, 50-150 mM MgCl2, and 15-28% Polyethylene glycol 4000 |
-Data collection
| Diffraction | Mean temperature: 113.15 K / Ambient temp details: Liquid Nitrogen Gas Stream |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRL SSRL  / Beamline: BL12-2 / Wavelength: 0.9795 Å / Beamline: BL12-2 / Wavelength: 0.9795 Å |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Feb 15, 2015 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9795 Å / Relative weight: 1 |
| Reflection | Resolution: 3→58.98 Å / Num. obs: 31728 / % possible obs: 94.88 % / Redundancy: 3.1 % / Biso Wilson estimate: 65.6 Å2 / CC1/2: 0.914 / Rmerge(I) obs: 0.1026 / Net I/σ(I): 6.66 |
| Reflection shell | Resolution: 3→3.107 Å / Redundancy: 2.8 % / Rmerge(I) obs: 0.6214 / Mean I/σ(I) obs: 1.37 / % possible all: 90.53 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5JA1 Resolution: 3→58.974 Å / SU ML: 0.37 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 24.25 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3→58.974 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj