[English] 日本語
 Yorodumi
Yorodumi- PDB-5if5: Crystal structure of polymerase acid protein (PA) from Influenza ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5if5 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of polymerase acid protein (PA) from Influenza A virus, WILSON-SMITH/1933 (H1N1) bound to EBSI-39 (2,3-dichlorophenyl)methanol | ||||||
 Components Components | Polymerase acidic protein | ||||||
 Keywords Keywords | VIRAL PROTEIN / NIAID / structural genomics / flu / fragment screening / Seattle Structural Genomics Center for Infectious Disease / SSGCID | ||||||
| Function / homology |  Function and homology information Function and homology informationRNA polymerase activity / negative regulation of viral transcription / negative stranded viral RNA replication / negative regulation of viral genome replication / cap snatching / symbiont-mediated suppression of host mRNA transcription via inhibition of RNA polymerase II activity / protein import into nucleus / endonuclease activity / Hydrolases; Acting on ester bonds / host cell cytoplasm ...RNA polymerase activity / negative regulation of viral transcription / negative stranded viral RNA replication / negative regulation of viral genome replication / cap snatching / symbiont-mediated suppression of host mRNA transcription via inhibition of RNA polymerase II activity / protein import into nucleus / endonuclease activity / Hydrolases; Acting on ester bonds / host cell cytoplasm / symbiont-mediated suppression of host gene expression / viral translational frameshifting / DNA-templated transcription / host cell nucleus / RNA binding / metal ion binding Similarity search - Function | ||||||
| Biological species |   Influenza A virus Influenza A virus | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 2.25 Å molecular replacement / Resolution: 2.25 Å | ||||||
 Authors Authors | Seattle Structural Genomics Center for Infectious Disease (SSGCID) | ||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Fragment screening by STD NMR identifies novel site binders against influenza A virus polymerase PA Authors: Pierce, P. / Muruthi, M.M. / Abendroth, J. / Moen, S.O. / Begley, D.W. / Davies, D.R. / Marathias, V.M. / Staker, B.L. / Myler, P.J. / Lorimer, D.D. / Edwards, T.E. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5if5.cif.gz 5if5.cif.gz | 181 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5if5.ent.gz pdb5if5.ent.gz | 142.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5if5.json.gz 5if5.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  5if5_validation.pdf.gz 5if5_validation.pdf.gz | 463.6 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  5if5_full_validation.pdf.gz 5if5_full_validation.pdf.gz | 465.4 KB | Display | |
| Data in XML |  5if5_validation.xml.gz 5if5_validation.xml.gz | 17.8 KB | Display | |
| Data in CIF |  5if5_validation.cif.gz 5if5_validation.cif.gz | 25.6 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/if/5if5 https://data.pdbj.org/pub/pdb/validation_reports/if/5if5 ftp://data.pdbj.org/pub/pdb/validation_reports/if/5if5 ftp://data.pdbj.org/pub/pdb/validation_reports/if/5if5 | HTTPS FTP |
-Related structure data
| Related structure data |  5ieqC  5if2C  5if7C  5if8C  5ifbC  5ifcC  5ifdC C: citing same article ( |
|---|---|
| Similar structure data | |
| Other databases |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 53096.723 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Influenza A virus (strain A/Wilson-Smith/1933 H1N1) Influenza A virus (strain A/Wilson-Smith/1933 H1N1)Strain: A/Wilson-Smith/1933 H1N1 / Gene: PA / Production host:  | ||||||
|---|---|---|---|---|---|---|---|
| #2: Chemical | ChemComp-DMS / | ||||||
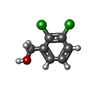
| #3: Chemical | | #4: Chemical | ChemComp-6B0 / ( | #5: Water | ChemComp-HOH / | Compound details | The distance between Residue A GLU 677 and Residue A PHE 681 is 20.95 Angstrom despite the fact ...The distance between Residue A GLU 677 and Residue A PHE 681 is 20.95 Angstrom despite the fact that there are only 3 residues (not enough sequence) to cover the gap region. One possibility for that is some domain swapping for the last two helices (note that E677 is closer to the symmetry related F681). Another possibility is that the protein is clipped. There is a bit of a doublet in the SDS PAGE of this protein. | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.52 Å3/Da / Density % sol: 51.16 % |
|---|---|
| Crystal grow | Temperature: 289 K / Method: vapor diffusion, sitting drop / pH: 8.5 Details: 20 mg/mL protein against Morpheus D10 with 10% PEG 8000, 20% EG, 0.02 M each alcohol, 0.1 M bicine/Trisma base pH 8.5, crystal tracking ID 263964d10, unique puck ID pyl5-5 |
-Data collection
| Diffraction | Mean temperature: 100 K | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 21-ID-F / Wavelength: 0.97872 Å / Beamline: 21-ID-F / Wavelength: 0.97872 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: MARMOSAIC 300 mm CCD / Detector: CCD / Date: Aug 13, 2014 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.97872 Å / Relative weight: 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.25→50 Å / Num. obs: 27497 / % possible obs: 99.9 % / Observed criterion σ(I): -3 / Redundancy: 5.2 % / Biso Wilson estimate: 35.2 Å2 / CC1/2: 0.995 / Rmerge(I) obs: 0.101 / Net I/σ(I): 12.91 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell |
|
-Phasing
| Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT / Resolution: 2.25→47.436 Å / SU ML: 0.22 / Cross valid method: FREE R-VALUE / σ(F): 1.37 / Phase error: 18.57 / Stereochemistry target values: ML MOLECULAR REPLACEMENT / Resolution: 2.25→47.436 Å / SU ML: 0.22 / Cross valid method: FREE R-VALUE / σ(F): 1.37 / Phase error: 18.57 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.25→47.436 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller














 PDBj
PDBj