[English] 日本語
 Yorodumi
Yorodumi- PDB-4zvr: Caspase-7 Variant 4 (V4) with reprogrammed substrate specificity ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4zvr | ||||||
|---|---|---|---|---|---|---|---|
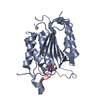
| Title | Caspase-7 Variant 4 (V4) with reprogrammed substrate specificity due to Y230V/W232Y/S234V/Q276D substitutions bound to DEVD inhibitor. | ||||||
 Components Components |
| ||||||
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR / Directed Evolution / Protease / Peptide Inhibitor / Designed Active Site Specificity / HYDROLASE-HYDROLASE INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationlymphocyte apoptotic process / caspase-7 / positive regulation of plasma membrane repair / cellular response to staurosporine / SMAC, XIAP-regulated apoptotic response / plasma membrane repair / Activation of caspases through apoptosome-mediated cleavage / SMAC (DIABLO) binds to IAPs / SMAC(DIABLO)-mediated dissociation of IAP:caspase complexes / fibroblast apoptotic process ...lymphocyte apoptotic process / caspase-7 / positive regulation of plasma membrane repair / cellular response to staurosporine / SMAC, XIAP-regulated apoptotic response / plasma membrane repair / Activation of caspases through apoptosome-mediated cleavage / SMAC (DIABLO) binds to IAPs / SMAC(DIABLO)-mediated dissociation of IAP:caspase complexes / fibroblast apoptotic process / ceramide biosynthetic process / execution phase of apoptosis / Apoptotic cleavage of cellular proteins / Caspase-mediated cleavage of cytoskeletal proteins / response to UV / striated muscle cell differentiation / cysteine-type peptidase activity / protein catabolic process / protein maturation / protein processing / fibrillar center / peptidase activity / positive regulation of neuron apoptotic process / heart development / cellular response to lipopolysaccharide / neuron apoptotic process / aspartic-type endopeptidase activity / defense response to bacterium / cysteine-type endopeptidase activity / apoptotic process / proteolysis / : / RNA binding / nucleoplasm / nucleus / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human)synthetic construct (others) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.3 Å MOLECULAR REPLACEMENT / Resolution: 2.3 Å | ||||||
 Authors Authors | Hill, M.E. / MacPherson, D.J. / Hardy, J.A. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Acs Chem.Biol. / Year: 2016 Journal: Acs Chem.Biol. / Year: 2016Title: Reprogramming Caspase-7 Specificity by Regio-Specific Mutations and Selection Provides Alternate Solutions for Substrate Recognition. Authors: Hill, M.E. / MacPherson, D.J. / Wu, P. / Julien, O. / Wells, J.A. / Hardy, J.A. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4zvr.cif.gz 4zvr.cif.gz | 113.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4zvr.ent.gz pdb4zvr.ent.gz | 84.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4zvr.json.gz 4zvr.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/zv/4zvr https://data.pdbj.org/pub/pdb/validation_reports/zv/4zvr ftp://data.pdbj.org/pub/pdb/validation_reports/zv/4zvr ftp://data.pdbj.org/pub/pdb/validation_reports/zv/4zvr | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4zvoC  4zvpC  4zvqC  4zvsC  4zvtC  4zvuC  3edrS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 22189.203 Da / Num. of mol.: 2 / Fragment: UNP residues 34-231 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CASP7, MCH3 / Production host: Homo sapiens (human) / Gene: CASP7, MCH3 / Production host:  #2: Protein | Mass: 13135.732 Da / Num. of mol.: 2 / Fragment: UNP residues 232-336 / Mutation: Y230V,W232Y,S234V,Q276D Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CASP7, MCH3 / Production host: Homo sapiens (human) / Gene: CASP7, MCH3 / Production host:  #3: Protein/peptide | #4: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.01 Å3/Da / Density % sol: 59.16 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, hanging drop / pH: 5 Details: 300 mM diammonium citrate, 14% PEG 3350, 10 mM GuHCl, 20% glycerol |
-Data collection
| Diffraction | Mean temperature: 100 K | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 24-ID-E / Wavelength: 0.97919 Å / Beamline: 24-ID-E / Wavelength: 0.97919 Å | |||||||||||||||||||||||||||
| Detector | Type: ADSC QUANTUM 315 / Detector: CCD / Date: Oct 29, 2014 | |||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.97919 Å / Relative weight: 1 | |||||||||||||||||||||||||||
| Reflection | Resolution: 2.3→76.19 Å / Num. obs: 38059 / % possible obs: 99.9 % / Redundancy: 7.4 % / CC1/2: 0.998 / Rpim(I) all: 0.043 / Rsym value: 0.104 / Net I/σ(I): 12.3 / Num. measured all: 282011 / Scaling rejects: 14 | |||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1 / Rejects: _
|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 3EDR Resolution: 2.3→70.577 Å / SU ML: 0.27 / Cross valid method: FREE R-VALUE / σ(F): 0.21 / Phase error: 24.67 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.3→70.577 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller















 PDBj
PDBj