[English] 日本語
 Yorodumi
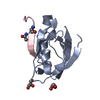
Yorodumi- PDB-4jog: CFTR Associated Ligand (CAL) PDZ domain bound to peptide V-iCAL36... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4jog | ||||||
|---|---|---|---|---|---|---|---|
| Title | CFTR Associated Ligand (CAL) PDZ domain bound to peptide V-iCAL36 (ANSRVPTSII) | ||||||
 Components Components |
| ||||||
 Keywords Keywords | PEPTIDE BINDING PROTEIN / PDZ / CFTR Associated Ligand / CAL / PIST / FIG / PDZ-peptide complex | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of anion channel activity / RHO GTPases regulate CFTR trafficking / negative regulation of protein localization to cell surface / Golgi to plasma membrane transport / trans-Golgi network transport vesicle / Golgi-associated vesicle membrane / apical protein localization / RHOQ GTPase cycle / molecular sequestering activity / endoplasmic reticulum to Golgi vesicle-mediated transport ...negative regulation of anion channel activity / RHO GTPases regulate CFTR trafficking / negative regulation of protein localization to cell surface / Golgi to plasma membrane transport / trans-Golgi network transport vesicle / Golgi-associated vesicle membrane / apical protein localization / RHOQ GTPase cycle / molecular sequestering activity / endoplasmic reticulum to Golgi vesicle-mediated transport / protein transport / transmembrane transporter binding / postsynaptic density / Golgi membrane / lysosomal membrane / dendrite / Golgi apparatus / protein-containing complex / membrane / identical protein binding / plasma membrane / cytoplasm Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.465 Å MOLECULAR REPLACEMENT / Resolution: 1.465 Å | ||||||
 Authors Authors | Amacher, J.F. / Madden, D.R. | ||||||
 Citation Citation |  Journal: Structure / Year: 2014 Journal: Structure / Year: 2014Title: Stereochemical Preferences Modulate Affinity and Selectivity among Five PDZ Domains that Bind CFTR: Comparative Structural and Sequence Analyses. Authors: Amacher, J.F. / Cushing, P.R. / Brooks, L. / Boisguerin, P. / Madden, D.R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4jog.cif.gz 4jog.cif.gz | 85 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4jog.ent.gz pdb4jog.ent.gz | 64.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4jog.json.gz 4jog.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/jo/4jog https://data.pdbj.org/pub/pdb/validation_reports/jo/4jog ftp://data.pdbj.org/pub/pdb/validation_reports/jo/4jog ftp://data.pdbj.org/pub/pdb/validation_reports/jo/4jog | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4joeC  4jofC  4johC  4jojC  4jokC  4jopC  4jorC  4k6yC  4k72C  4k75C  4k76C  4k78C  4e34S C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 9353.722 Da / Num. of mol.: 2 / Fragment: CAL PDZ domain Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GOPC, CAL, FIG / Plasmid: pET16b / Production host: Homo sapiens (human) / Gene: GOPC, CAL, FIG / Plasmid: pET16b / Production host:  #2: Protein/peptide | Mass: 1058.210 Da / Num. of mol.: 2 / Source method: obtained synthetically #3: Chemical | #4: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.07 Å3/Da / Density % sol: 40.44 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, hanging drop / pH: 7.5 Details: 40% (w/v) polyethylene glycol (PEG), 0.2 M sodium chloride, 0.1 M tris(hydroxymethyl)aminomethane (Tris), pH 7.5, VAPOR DIFFUSION, HANGING DROP, temperature 291K |
-Data collection
| Diffraction | Mean temperature: 100 K | ||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  NSLS NSLS  / Beamline: X6A / Wavelength: 1 Å / Beamline: X6A / Wavelength: 1 Å | ||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: ADSC QUANTUM 270 / Detector: CCD / Date: Nov 27, 2010 | ||||||||||||||||||||||||||||||||||||||||||
| Radiation | Monochromator: S1 111 CHANNEL / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 1.465→19.656 Å / Num. all: 30756 / Num. obs: 30171 / % possible obs: 98.1 % / Observed criterion σ(F): 1.99 / Observed criterion σ(I): 3.05 / Rmerge(I) obs: 0.088 / Rsym value: 0.068 / Net I/σ(I): 16.71 | ||||||||||||||||||||||||||||||||||||||||||
| Reflection shell |
|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB entry 4E34 (CAL PDZ domain bound to iCAL36 peptide) Resolution: 1.465→19.655 Å / SU ML: 0.18 / Cross valid method: Omit map / σ(F): 1.99 / σ(I): 3.05 / Phase error: 17.23 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.72 Å / VDW probe radii: 1 Å / Solvent model: FLAT BULK SOLVENT MODEL / Bsol: 38.287 Å2 / ksol: 0.413 e/Å3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.465→19.655 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj



