[English] 日本語
 Yorodumi
Yorodumi- PDB-4bvh: CRYSTAL STRUCTURE OF HUMAN SIRT3 IN COMPLEX WITH THE INHIBITOR EX... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4bvh | ||||||
|---|---|---|---|---|---|---|---|
| Title | CRYSTAL STRUCTURE OF HUMAN SIRT3 IN COMPLEX WITH THE INHIBITOR EX-527 AND 2'-O-ACETYL-ADP-RIBOSE | ||||||
 Components Components | NAD-DEPENDENT PROTEIN DEACETYLASE SIRTUIN-3, MITOCHONDRIAL | ||||||
 Keywords Keywords | HYDROLASE / INHIBITOR COMPLEX / EX-527 | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of superoxide dismutase activity / positive regulation of catalase activity / NAD-dependent protein lysine delactylase activity / positive regulation of ceramide biosynthetic process / peptidyl-lysine deacetylation / Maturation of TCA enzymes and regulation of TCA cycle / protein acetyllysine N-acetyltransferase / protein deacetylation / NAD-dependent protein lysine deacetylase activity / histone deacetylase activity, NAD-dependent ...positive regulation of superoxide dismutase activity / positive regulation of catalase activity / NAD-dependent protein lysine delactylase activity / positive regulation of ceramide biosynthetic process / peptidyl-lysine deacetylation / Maturation of TCA enzymes and regulation of TCA cycle / protein acetyllysine N-acetyltransferase / protein deacetylation / NAD-dependent protein lysine deacetylase activity / histone deacetylase activity, NAD-dependent / positive regulation of oxidative phosphorylation / Regulation of FOXO transcriptional activity by acetylation / protein lysine deacetylase activity / cellular response to stress / negative regulation of reactive oxygen species metabolic process / NAD+ binding / Mitochondrial unfolded protein response (UPRmt) / FOXO-mediated transcription of oxidative stress, metabolic and neuronal genes / Transferases; Acyltransferases; Transferring groups other than aminoacyl groups / aerobic respiration / Transcriptional activation of mitochondrial biogenesis / negative regulation of ERK1 and ERK2 cascade / positive regulation of insulin secretion / sequence-specific DNA binding / mitochondrial matrix / enzyme binding / protein-containing complex / mitochondrion / zinc ion binding / nucleoplasm / nucleus Similarity search - Function | ||||||
| Biological species |  HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.9 Å MOLECULAR REPLACEMENT / Resolution: 1.9 Å | ||||||
 Authors Authors | Gertz, M. / Weyand, M. / Steegborn, C. | ||||||
 Citation Citation |  Journal: Proc.Natl.Acad.Sci.USA / Year: 2013 Journal: Proc.Natl.Acad.Sci.USA / Year: 2013Title: Ex-527 Inhibits Sirtuins by Exploiting Their Unique Nad+-Dependent Deacetylation Mechanism Authors: Gertz, M. / Fischer, F. / Nguyen, G.T.T. / Lakshminarasimhan, M. / Schutkowski, M. / Weyand, M. / Steegborn, C. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4bvh.cif.gz 4bvh.cif.gz | 192.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4bvh.ent.gz pdb4bvh.ent.gz | 150.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4bvh.json.gz 4bvh.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/bv/4bvh https://data.pdbj.org/pub/pdb/validation_reports/bv/4bvh ftp://data.pdbj.org/pub/pdb/validation_reports/bv/4bvh ftp://data.pdbj.org/pub/pdb/validation_reports/bv/4bvh | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4buzC  4bv2C  4bv3C  4bvbC  4bveC  4bvfC  4bvgC  3gltS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||
| 2 | 
| ||||||||||||
| 3 | 
| ||||||||||||
| Unit cell |
| ||||||||||||
| Noncrystallographic symmetry (NCS) | NCS oper:
|
- Components
Components
-Protein , 1 types, 3 molecules ABC
| #1: Protein | Mass: 31484.219 Da / Num. of mol.: 3 / Fragment: RESIDUES 116-399 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  HOMO SAPIENS (human) / Plasmid: PVFT3S / Production host: HOMO SAPIENS (human) / Plasmid: PVFT3S / Production host:  References: UniProt: Q9NTG7, Hydrolases; Acting on carbon-nitrogen bonds, other than peptide bonds; In linear amides |
|---|
-Non-polymers , 9 types, 506 molecules 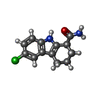
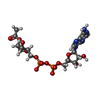



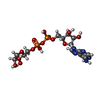











| #2: Chemical | | #3: Chemical | #4: Chemical | #5: Chemical | ChemComp-NA / | #6: Chemical | #7: Chemical | ChemComp-AR6 / [( | #8: Chemical | ChemComp-EDO / #9: Chemical | ChemComp-GOL / | #10: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.26 Å3/Da / Density % sol: 45.52 % / Description: NONE |
|---|---|
| Crystal grow | pH: 7 / Details: 20% PEG 6000, 0.1 M HEPES PH7 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  BESSY BESSY  / Beamline: 14.1 / Wavelength: 0.918 / Beamline: 14.1 / Wavelength: 0.918 |
| Detector | Type: MARMOSAIC 225 mm CCD / Detector: CCD / Date: May 30, 2012 / Details: COLLIMATOR |
| Radiation | Monochromator: SI(111) MONOCHROMATOR / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.918 Å / Relative weight: 1 |
| Reflection | Resolution: 1.9→34.4 Å / Num. obs: 68289 / % possible obs: 99.8 % / Observed criterion σ(I): -3 / Redundancy: 4.7 % / Rmerge(I) obs: 0.08 / Net I/σ(I): 16.3 |
| Reflection shell | Resolution: 1.9→1.95 Å / Redundancy: 4.8 % / Rmerge(I) obs: 0.49 / Mean I/σ(I) obs: 3.5 / % possible all: 100 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 3GLT Resolution: 1.9→34.44 Å / Cor.coef. Fo:Fc: 0.959 / Cor.coef. Fo:Fc free: 0.927 / SU B: 3.076 / SU ML: 0.092 / Cross valid method: THROUGHOUT / ESU R: 0.147 / ESU R Free: 0.145 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS. HYDROGENS HAVE BEEN USED IF PRESENT IN THE INPUT. U VALUES REFINED INDIVIDUALLY
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 27.027 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.9→34.44 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj


















