[English] 日本語
 Yorodumi
Yorodumi- PDB-3e3b: Crystal structure of catalytic subunit of human protein kinase CK... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3e3b | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of catalytic subunit of human protein kinase CK2alpha prime with a potent indazole-derivative inhibitor | ||||||
 Components Components | Casein kinase II subunit alpha' | ||||||
 Keywords Keywords | TRANSFERASE / Casein Kinase 2 / CK2alpha prime / CK2alpha inhibitor / selective kinase inhibitor / ATP-binding / Kinase / Nucleotide-binding / Polymorphism / Serine/threonine-protein kinase / Wnt signaling pathway | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of mitophagy / regulation of chromosome separation / WNT mediated activation of DVL / Condensation of Prometaphase Chromosomes / protein kinase CK2 complex / : / Phosphorylation and nuclear translocation of the CRY:PER:kinase complex / Regulation of CDH1 posttranslational processing and trafficking to plasma membrane / Receptor Mediated Mitophagy / Synthesis of PC ...regulation of mitophagy / regulation of chromosome separation / WNT mediated activation of DVL / Condensation of Prometaphase Chromosomes / protein kinase CK2 complex / : / Phosphorylation and nuclear translocation of the CRY:PER:kinase complex / Regulation of CDH1 posttranslational processing and trafficking to plasma membrane / Receptor Mediated Mitophagy / Synthesis of PC / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known / Maturation of hRSV A proteins / negative regulation of apoptotic signaling pathway / negative regulation of proteasomal ubiquitin-dependent protein catabolic process / liver regeneration / acrosomal vesicle / Signal transduction by L1 / cerebral cortex development / Wnt signaling pathway / Regulation of PTEN stability and activity / KEAP1-NFE2L2 pathway / double-strand break repair / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / spermatogenesis / Regulation of TP53 Activity through Phosphorylation / non-specific serine/threonine protein kinase / protein serine kinase activity / protein serine/threonine kinase activity / apoptotic process / DNA damage response / chromatin / nucleoplasm / ATP binding / nucleus / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3.2 Å MOLECULAR REPLACEMENT / Resolution: 3.2 Å | ||||||
 Authors Authors | Kinoshita, T. / Nakaniwa, T. / Tada, T. | ||||||
 Citation Citation |  Journal: Acta Crystallogr.,Sect.F / Year: 2009 Journal: Acta Crystallogr.,Sect.F / Year: 2009Title: Structure of human protein kinase CK2alpha2 with a potent indazole-derivative inhibitor Authors: Nakaniwa, T. / Kinoshita, T. / Sekiguchi, Y. / Tada, T. / Nakanishi, I. / Kitaura, K. / Suzuki, Y. / Ohno, H. / Hirasawa, A. / Tsujimoto, G. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3e3b.cif.gz 3e3b.cif.gz | 85.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3e3b.ent.gz pdb3e3b.ent.gz | 63 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3e3b.json.gz 3e3b.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/e3/3e3b https://data.pdbj.org/pub/pdb/validation_reports/e3/3e3b ftp://data.pdbj.org/pub/pdb/validation_reports/e3/3e3b ftp://data.pdbj.org/pub/pdb/validation_reports/e3/3e3b | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1plkS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 40159.984 Da / Num. of mol.: 1 / Fragment: residues in database 1-334 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CSNK2A2, CK2A2 / Plasmid: pGEX-6P-1 / Production host: Homo sapiens (human) / Gene: CSNK2A2, CK2A2 / Plasmid: pGEX-6P-1 / Production host:  References: UniProt: P19784, non-specific serine/threonine protein kinase |
|---|---|
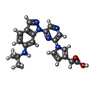
| #2: Chemical | ChemComp-CCK / [ |
| #3: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.01 Å3/Da / Density % sol: 38.9 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, sitting drop / pH: 8 Details: 8% PEG8000, 0.1M Tris-HCl, pH 8.0, VAPOR DIFFUSION, SITTING DROP, temperature 277K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Photon Factory Photon Factory  / Beamline: BL-6A / Beamline: BL-6A |
| Detector | Type: ADSC QUANTUM 4 / Detector: CCD / Date: May 18, 2007 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Relative weight: 1 |
| Reflection | Resolution: 3.2→57.64 Å / Num. obs: 5978 / % possible obs: 100 % / Observed criterion σ(F): 0 |
| Reflection shell | Resolution: 3.2→3.31 Å / % possible all: 100 |
- Processing
Processing
| Software |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1PLK Resolution: 3.2→57.64 Å / σ(F): 0 / Stereochemistry target values: Engh & Huber
| ||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3.2→57.64 Å
| ||||||||||||||||||||
| Refine LS restraints | Type: c_bond_d / Dev ideal: 0.008 |
 Movie
Movie Controller
Controller






















 PDBj
PDBj