[English] 日本語
 Yorodumi
Yorodumi- PDB-3dtu: Catalytic core subunits (I and II) of cytochrome c oxidase from R... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3dtu | ||||||
|---|---|---|---|---|---|---|---|
| Title | Catalytic core subunits (I and II) of cytochrome c oxidase from Rhodobacter sphaeroides complexed with deoxycholic acid | ||||||
 Components Components | (Cytochrome c oxidase subunit ...) x 2 | ||||||
 Keywords Keywords | OXIDOREDUCTASE / TRANSMEMBRANE PROTEIN COMPLEX / deoxycholic acid / Copper / Electron transport / Heme / Hydrogen ion transport / Ion transport / Iron / Membrane / Metal-binding / Respiratory chain / Transmembrane / Transport | ||||||
| Function / homology |  Function and homology information Function and homology informationrespiratory chain complex IV / cytochrome-c oxidase / oxidative phosphorylation / cytochrome-c oxidase activity / electron transport coupled proton transport / ATP synthesis coupled electron transport / respiratory electron transport chain / oxidoreductase activity / copper ion binding / heme binding ...respiratory chain complex IV / cytochrome-c oxidase / oxidative phosphorylation / cytochrome-c oxidase activity / electron transport coupled proton transport / ATP synthesis coupled electron transport / respiratory electron transport chain / oxidoreductase activity / copper ion binding / heme binding / metal ion binding / plasma membrane Similarity search - Function | ||||||
| Biological species |  Rhodobacter sphaeroides (bacteria) Rhodobacter sphaeroides (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / Resolution: 2.15 Å SYNCHROTRON / Resolution: 2.15 Å | ||||||
 Authors Authors | Qin, L. / Mills, D.A. / Buhrow, L. / Hiser, C. / Ferguson-Miller, S. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 2008 Journal: Biochemistry / Year: 2008Title: A conserved steroid binding site in cytochrome C oxidase. Authors: Qin, L. / Mills, D.A. / Buhrow, L. / Hiser, C. / Ferguson-Miller, S. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3dtu.cif.gz 3dtu.cif.gz | 362.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3dtu.ent.gz pdb3dtu.ent.gz | 288.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3dtu.json.gz 3dtu.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/dt/3dtu https://data.pdbj.org/pub/pdb/validation_reports/dt/3dtu ftp://data.pdbj.org/pub/pdb/validation_reports/dt/3dtu ftp://data.pdbj.org/pub/pdb/validation_reports/dt/3dtu | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2gsmS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Cytochrome c oxidase subunit ... , 2 types, 4 molecules ACBD
| #1: Protein | Mass: 63195.383 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Rhodobacter sphaeroides (bacteria) / Strain: 37delta4 / Gene: ctaD / Plasmid: PRK415 / Production host: Rhodobacter sphaeroides (bacteria) / Strain: 37delta4 / Gene: ctaD / Plasmid: PRK415 / Production host:  Rhodobacter sphaeroides (bacteria) / Strain (production host): deltaIdeltaIV / References: UniProt: P33517, cytochrome-c oxidase Rhodobacter sphaeroides (bacteria) / Strain (production host): deltaIdeltaIV / References: UniProt: P33517, cytochrome-c oxidase#2: Protein | Mass: 29365.385 Da / Num. of mol.: 2 / Fragment: UNP residues 26-281 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Rhodobacter sphaeroides (bacteria) / Strain: 37delta4 / Gene: ctaC, coxII, ctaB / Plasmid: PRK415 / Production host: Rhodobacter sphaeroides (bacteria) / Strain: 37delta4 / Gene: ctaC, coxII, ctaB / Plasmid: PRK415 / Production host:  Rhodobacter sphaeroides (bacteria) / Strain (production host): deltaIdeltaIV / References: UniProt: Q03736, cytochrome-c oxidase Rhodobacter sphaeroides (bacteria) / Strain (production host): deltaIdeltaIV / References: UniProt: Q03736, cytochrome-c oxidase |
|---|
-Sugars , 1 types, 9 molecules 
| #3: Sugar | ChemComp-DMU / |
|---|
-Non-polymers , 11 types, 684 molecules 







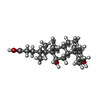











| #4: Chemical | ChemComp-CU / #5: Chemical | #6: Chemical | #7: Chemical | #8: Chemical | #9: Chemical | ChemComp-HEA / #10: Chemical | ChemComp-TRD / #11: Chemical | ChemComp-CD / #12: Chemical | ChemComp-HTO / | #13: Chemical | ChemComp-DXC / ( | #14: Water | ChemComp-HOH / | |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.69 Å3/Da / Density % sol: 66.68 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, sitting drop / pH: 6.3 Details: 24% PEG-400, 5mM deoxycholic acid , pH 6.3, VAPOR DIFFUSION, SITTING DROP, temperature 277K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 21-ID-G / Wavelength: 0.9785 Å / Beamline: 21-ID-G / Wavelength: 0.9785 Å |
| Detector | Type: MARMOSAIC 300 mm CCD / Detector: CCD / Date: Nov 15, 2007 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9785 Å / Relative weight: 1 |
| Reflection | Resolution: 2.15→50 Å / Num. all: 150058 / Num. obs: 145257 / % possible obs: 96.8 % / Redundancy: 6.6 % / Rmerge(I) obs: 0.069 / Net I/σ(I): 21.9 |
| Reflection shell | Resolution: 2.15→2.33 Å / Redundancy: 3.8 % / Rmerge(I) obs: 0.513 / Mean I/σ(I) obs: 2.1 / Num. unique all: 11510 / % possible all: 77.6 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Starting model: PDB entry 2GSM Resolution: 2.15→40 Å / Cor.coef. Fo:Fc: 0.953 / Cor.coef. Fo:Fc free: 0.942 / Occupancy max: 1 / Occupancy min: 0 / SU B: 3.842 / SU ML: 0.101 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.164 / ESU R Free: 0.146 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: (1) THERE IS A COVALENT LINKAGE BETWEEN NE2 OF HIS284 OF SUBUNIT I AND CE2 OF TYR288 OF SUBUNIT I. (2) THERE IS RESIDUAL DENSITY IN (FO-FC) DIFFERENCE FOURIER MAP AT THE MAGNESIUM SITES IN ...Details: (1) THERE IS A COVALENT LINKAGE BETWEEN NE2 OF HIS284 OF SUBUNIT I AND CE2 OF TYR288 OF SUBUNIT I. (2) THERE IS RESIDUAL DENSITY IN (FO-FC) DIFFERENCE FOURIER MAP AT THE MAGNESIUM SITES IN BOTH MOLECULES AND THE B-FACTOR FOR EACH MAGNESIUM ION IS UNUSUALLY LOW. THESE OBSERVATIONS SUGGEST THAT THIS MAGNESIUM SITE MAY BE PARTIALLY OCCUPIED BY A HEAVIER METAL ION IN THE PROTEIN CRYSTAL. (3) THE SIDE CHAINS OF THE FOLLOWING RESIDUES ARE NOT FULLY RESOLVED. THE UNRESOLVED SIDE CHAIN ATOMS ARE ASSIGNED AN OCCUPANCY OF 0.00 AND AN ARBITORY B-FACTOR OF 100.00: ARG-A-13 LYS-A-74 PHE-A-77 ARG-A-137 LYS-A-224 GLU-A-533 GLU-A-548 LYS-B-86 ARG-B-93 GLU-B-131 GLU-B-182 ARG-B-187 PHE-C-17 THR-C-18 ARG-C-19 GLU-C-69 LYS-C-74 ARG-C-521 GLU-C-533 LYS-D-86 LYS-D-89 GLU-D-131 HIS-D-284 (4) LIGANDS LABELLED AS TRD ARE ALL ALKYL CHAINS (WITH DIFFERENT LENGTHS) OF EITHER DMU OR NATIVE MEMBRANE LIPIDS SUCH AS PHOSPHATIDYL ETHANOLAMINE OR CARDIOLIPIN. SOME OF THE DETERGENTS LABELLED AS DMU DO NOT CONTAIN THE CARBON CHAIN, OR THE DISTAL GLUCOSE MOIETY. THE AUTHORS DO NOT KNOW FOR SURE THE IDENTITIES OF THE COMPLETE MOLECULES YET.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 100 Å2 / Biso mean: 38.516 Å2 / Biso min: 15.16 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.15→40 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.15→2.206 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller












 PDBj
PDBj

















