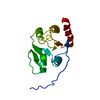
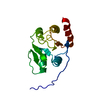
Entry Database : PDB / ID : 2rnkTitle NMR structure of the domain 513-651 of the SARS-CoV nonstructural protein nsp3 Replicase polyprotein 1ab Keywords / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Method / Model details The middle domain of the SARS-unique region of the nonstructural protein 3 of the SARS coronavirus Authors Chatterjee, A. / Johnson, M.A. / Serrano, P. / Pedrini, B. / Joseph, J. / Saikatendu, K. / Neuman, B.W. / Wilson, I.A. / Stevens, R.C. / Buchmeier, M.J. ...Chatterjee, A. / Johnson, M.A. / Serrano, P. / Pedrini, B. / Joseph, J. / Saikatendu, K. / Neuman, B.W. / Wilson, I.A. / Stevens, R.C. / Buchmeier, M.J. / Kuhn, P. / Wuthrich, K. / Joint Center for Structural Genomics (JCSG) Journal : J.Virol. / Year : 2009Title : Nuclear magnetic resonance structure shows that the severe acute respiratory syndrome coronavirus-unique domain contains a macrodomain fold.Authors : Chatterjee, A. / Johnson, M.A. / Serrano, P. / Pedrini, B. / Joseph, J.S. / Neuman, B.W. / Saikatendu, K. / Buchmeier, M.J. / Kuhn, P. / Wuthrich, K. History Deposition Jan 11, 2008 Deposition site / Processing site Supersession Feb 5, 2008 ID 2JWI Revision 1.0 Feb 5, 2008 Provider / Type Revision 1.1 Jul 13, 2011 Group Revision 1.2 Mar 16, 2022 Group / Database references / Derived calculationsCategory database_2 / pdbx_nmr_spectrometer ... database_2 / pdbx_nmr_spectrometer / pdbx_struct_assembly / pdbx_struct_oper_list Item / _database_2.pdbx_database_accession / _pdbx_nmr_spectrometer.modelRevision 1.3 Feb 1, 2023 Group / Category / Item Revision 1.4 May 22, 2024 Group / Category / chem_comp_bond
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information SARS coronavirus
SARS coronavirus Authors
Authors Citation
Citation Journal: J.Virol. / Year: 2009
Journal: J.Virol. / Year: 2009 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 2rnk.cif.gz
2rnk.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb2rnk.ent.gz
pdb2rnk.ent.gz PDB format
PDB format 2rnk.json.gz
2rnk.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/rn/2rnk
https://data.pdbj.org/pub/pdb/validation_reports/rn/2rnk ftp://data.pdbj.org/pub/pdb/validation_reports/rn/2rnk
ftp://data.pdbj.org/pub/pdb/validation_reports/rn/2rnk

 Links
Links Assembly
Assembly
 Components
Components SARS coronavirus / Genus: Coronavirus / Gene: rep, 1a-1b / Plasmid: pET28b / Species (production host): Escherichia coli / Production host:
SARS coronavirus / Genus: Coronavirus / Gene: rep, 1a-1b / Plasmid: pET28b / Species (production host): Escherichia coli / Production host: 
 Sample preparation
Sample preparation Processing
Processing Movie
Movie Controller
Controller











 PDBj
PDBj







 HSQC
HSQC