[English] 日本語
 Yorodumi
Yorodumi- PDB-2o3z: X-ray crystal structure of LpxC complexed with 3-heptyloxybenzoate -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2o3z | ||||||
|---|---|---|---|---|---|---|---|
| Title | X-ray crystal structure of LpxC complexed with 3-heptyloxybenzoate | ||||||
 Components Components | UDP-3-O-[3-hydroxymyristoyl] N-acetylglucosamine deacetylase | ||||||
 Keywords Keywords | HYDROLASE / Lipid A biosynthesis / Lipid synthesis / LpxC / 3-heptyloxybenzoate | ||||||
| Function / homology |  Function and homology information Function and homology informationUDP-3-O-acyl-N-acetylglucosamine deacetylase / UDP-3-O-acyl-N-acetylglucosamine deacetylase activity / lipid A biosynthetic process / membrane / metal ion binding / cytoplasm Similarity search - Function | ||||||
| Biological species |   Aquifex aeolicus (bacteria) Aquifex aeolicus (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.25 Å MOLECULAR REPLACEMENT / Resolution: 2.25 Å | ||||||
 Authors Authors | Gennadios, H.A. / Whittington, D.A. / Christianson, D.W. | ||||||
 Citation Citation |  Journal: Bioorg.Med.Chem. / Year: 2007 Journal: Bioorg.Med.Chem. / Year: 2007Title: Amphipathic benzoic acid derivatives: synthesis and binding in the hydrophobic tunnel of the zinc deacetylase LpxC. Authors: Shin, H. / Gennadios, H.A. / Whittington, D.A. / Christianson, D.W. | ||||||
| History |
| ||||||
| Remark 999 | SEQUENCE THE RESIDUE NUMBERING SYSTEM FOLLOWS THAT OF THE E.COLI ENZYME, AND RESULTS IN A BREAKS IN ...SEQUENCE THE RESIDUE NUMBERING SYSTEM FOLLOWS THAT OF THE E.COLI ENZYME, AND RESULTS IN A BREAKS IN THE SEQUENTIAL NUMBERING IN FEW REGIONS |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2o3z.cif.gz 2o3z.cif.gz | 125.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2o3z.ent.gz pdb2o3z.ent.gz | 97.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2o3z.json.gz 2o3z.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/o3/2o3z https://data.pdbj.org/pub/pdb/validation_reports/o3/2o3z ftp://data.pdbj.org/pub/pdb/validation_reports/o3/2o3z ftp://data.pdbj.org/pub/pdb/validation_reports/o3/2o3z | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1p42S S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| 3 | 
| ||||||||
| 4 | 
| ||||||||
| Unit cell |
| ||||||||
| Details | There are 2 monomers in the asymmetric unit forming a crystallograpic dimer. The second monomer is generated by: X,Y,Z Y,X-Y,1/3+Z -X+Y,-X,2/3+Z -X,-Y,1/2+Z Y,-X+Y,5/6+Z X-Y,X,1/6+Z |
- Components
Components
-Protein , 1 types, 2 molecules AB
| #1: Protein | Mass: 30989.510 Da / Num. of mol.: 2 / Mutation: deltaD284-L294, C193A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Aquifex aeolicus (bacteria) / Gene: lpxC, envA / Plasmid: pET21a / Production host: Aquifex aeolicus (bacteria) / Gene: lpxC, envA / Plasmid: pET21a / Production host:  References: UniProt: O67648, Hydrolases; Acting on carbon-nitrogen bonds, other than peptide bonds; In linear amides |
|---|
-Non-polymers , 5 types, 178 molecules 


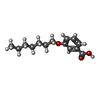





| #2: Chemical | | #3: Chemical | ChemComp-ZN / #4: Chemical | ChemComp-CL / | #5: Chemical | #6: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.91 Å3/Da / Density % sol: 57.79 % |
|---|---|
| Crystal grow | Temperature: 294 K / Method: vapor diffusion, hanging drop / pH: 7 Details: 100mM HEPES pH 7.0, 12-14% PEG3350, 180mM NaCl, 0.5mM ZnSO4, VAPOR DIFFUSION, HANGING DROP, temperature 294K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  NSLS NSLS  / Beamline: X29A / Wavelength: 0.9 Å / Beamline: X29A / Wavelength: 0.9 Å |
| Detector | Type: ADSC QUANTUM 315 / Detector: CCD / Date: Jun 6, 2005 Details: sagital focusing monochromator and vertical focusing mirror |
| Radiation | Monochromator: Rosenbaum-Rock double crystal / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9 Å / Relative weight: 1 |
| Reflection | Resolution: 2.25→50 Å / Num. obs: 33203 / % possible obs: 98.7 % / Redundancy: 2.8 % / Biso Wilson estimate: 24.6 Å2 / Net I/σ(I): 10.4 |
| Reflection shell | Resolution: 2.25→2.28 Å / % possible obs: 99.8 % / Redundancy: 2.6 % / Mean I/σ(I) obs: 2.5 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB entry 1P42 Resolution: 2.25→46.7 Å / Cross valid method: THROUGHOUT / Stereochemistry target values: Engh & Huber
| ||||||||||||||||||||||||||||
| Displacement parameters |
| ||||||||||||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.25 Å / Luzzati d res low obs: 5 Å / Luzzati sigma a obs: 0.21 Å | ||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.25→46.7 Å
| ||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj



