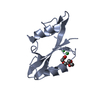
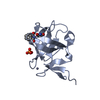
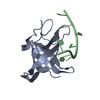
Entry Database : PDB / ID : 2mrjTitle Structure of Fyn protein SH2 bound Tyrosine-protein kinase Fyn Keywords / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Homo sapiens (human)Method / Model details closest to the average, model1 Authors Huculeci, R. / Buts, L. / Lenaerts, T. / Van Nuland, N. Journal : Structure / Year : 2016Title : Dynamically Coupled Residues within the SH2 Domain of FYN Are Key to Unlocking Its Activity.Authors : Huculeci, R. / Cilia, E. / Lyczek, A. / Buts, L. / Houben, K. / Seeliger, M.A. / van Nuland, N. / Lenaerts, T. History Deposition Jul 9, 2014 Deposition site / Processing site Revision 1.0 Jul 15, 2015 Provider / Type Revision 1.1 Apr 27, 2016 Group / Structure summaryRevision 1.2 Oct 26, 2016 Group Revision 1.3 Dec 14, 2016 Group Revision 1.4 Jun 14, 2023 Group / OtherCategory / pdbx_database_status / struct_ref_seq_difItem _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_database_status.status_code_nmr_data / _struct_ref_seq_dif.details Revision 1.5 May 15, 2024 Group / Database references / Category / chem_comp_bond / database_2 / Item
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Homo sapiens (human)
Homo sapiens (human) Authors
Authors Citation
Citation Journal: Structure / Year: 2016
Journal: Structure / Year: 2016 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 2mrj.cif.gz
2mrj.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb2mrj.ent.gz
pdb2mrj.ent.gz PDB format
PDB format 2mrj.json.gz
2mrj.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/mr/2mrj
https://data.pdbj.org/pub/pdb/validation_reports/mr/2mrj ftp://data.pdbj.org/pub/pdb/validation_reports/mr/2mrj
ftp://data.pdbj.org/pub/pdb/validation_reports/mr/2mrj

 Links
Links Assembly
Assembly
 Components
Components Homo sapiens (human) / Gene: FYN / Production host:
Homo sapiens (human) / Gene: FYN / Production host: 
 Sample preparation
Sample preparation Processing
Processing Movie
Movie Controller
Controller










 PDBj
PDBj



















 HSQC
HSQC