| NMR software | | 名称 | バージョン | 開発者 | 分類 |
|---|
| ARIA | 2.2 | Linge, O'Donoghue and Nilges| 構造決定 | | ARIA | 2.2 | Linge, O'Donoghue and Nilgesnoe assignment| CNS | 1.1 | Brunger, Adams, Clore, Gros, Nilges and Read| 構造決定 | | CNS | 1.1 | Brunger, Adams, Clore, Gros, Nilges and Read| 精密化 | | VnmrJ | 2.1BVariancollection NMRPipe NMRPipe | | Delaglio, Grzesiek, Vuister, Zhu, Pfeifer and Bax| 解析 | | NMRView | 5.2.2Johnson, One Moon Scientific| データ解析 | | NMRView | 5.2.2Johnson, One Moon Scientificchemical shift assignment| NMRView | 5.2.2Johnson, One Moon Scientificpeak picking| ProcheckNMR | 3.4 | Laskowski and MacArthur| データ解析 | | WHAT IF | | Vriend| データ解析 | | | | | | | | | | | | | | | | | | | |
|
|---|
| 精密化 | 手法: simulated annealing / ソフトェア番号: 1
詳細: Residues 1-12 are disordered. Only coordinates for residues 11-56 are included. Structures were calculated with restraints for residues 11-56. |
|---|
| NMR constraints | NOE constraints total: 4072 / NOE intraresidue total count: 1075 / NOE long range total count: 453 / NOE medium range total count: 740 / NOE sequential total count: 808 / Hydrogen bond constraints total count: 54 / Protein chi angle constraints total count: 0 / Protein other angle constraints total count: 0 / Protein phi angle constraints total count: 60 / Protein psi angle constraints total count: 0 |
|---|
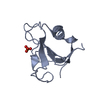
| 代表構造 | 選択基準: lowest energy |
|---|
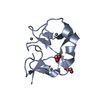
| NMRアンサンブル | コンフォーマー選択の基準: structures with the lowest energy
計算したコンフォーマーの数: 200 / 登録したコンフォーマーの数: 10 / Maximum lower distance constraint violation: 0.1 Å / Maximum torsion angle constraint violation: 5 ° / Maximum upper distance constraint violation: 0.1 Å / Torsion angle constraint violation method: CNS |
|---|
| NMR ensemble rms | Distance rms dev: 0.0205 Å / Distance rms dev error: 0.0001 Å |
|---|
 データを開く
データを開く 基本情報
基本情報 要素
要素 キーワード
キーワード 機能・相同性情報
機能・相同性情報
 Sulfolobus islandicus rod-shaped virus 1 (ウイルス)
Sulfolobus islandicus rod-shaped virus 1 (ウイルス) データ登録者
データ登録者 引用
引用 ジャーナル: J.Biol.Chem. / 年: 2009
ジャーナル: J.Biol.Chem. / 年: 2009 構造の表示
構造の表示 Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク ダウンロード
ダウンロード 2kel.cif.gz
2kel.cif.gz PDBx/mmCIF形式
PDBx/mmCIF形式 pdb2kel.ent.gz
pdb2kel.ent.gz PDB形式
PDB形式 2kel.json.gz
2kel.json.gz PDBx/mmJSON形式
PDBx/mmJSON形式 その他のダウンロード
その他のダウンロード https://data.pdbj.org/pub/pdb/validation_reports/ke/2kel
https://data.pdbj.org/pub/pdb/validation_reports/ke/2kel ftp://data.pdbj.org/pub/pdb/validation_reports/ke/2kel
ftp://data.pdbj.org/pub/pdb/validation_reports/ke/2kel リンク
リンク 集合体
集合体
 要素
要素
 Sulfolobus islandicus rod-shaped virus 1 (ウイルス)
Sulfolobus islandicus rod-shaped virus 1 (ウイルス)
 試料調製
試料調製 解析
解析 ムービー
ムービー コントローラー
コントローラー









 PDBj
PDBj NMRPipe
NMRPipe