登録情報 データベース : PDB / ID : 2dmvタイトル Solution structure of the second ww domain of Itchy homolog E3 ubiquitin protein ligase (Itch) Itchy homolog E3 ubiquitin protein ligase キーワード / / / / / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト)手法 / データ登録者 Ohnishi, S. / Paakkonen, K. / Tochio, N. / Tomizawa, T. / Koshiba, S. / Inoue, M. / Guntert, P. / Kigawa, T. / Yokoyama, S. / RIKEN Structural Genomics/Proteomics Initiative (RSGI) ジャーナル : To be Published タイトル : Solution structure of the second ww domain of Itchy homolog E3 ubiquitin protein ligase (Itch)著者 : Ohnishi, S. / Paakkonen, K. / Tochio, N. / Tomizawa, T. / Koshiba, S. / Inoue, M. / Guntert, P. / Kigawa, T. / Yokoyama, S. 履歴 登録 2006年4月24日 登録サイト / 処理サイト 改定 1.0 2006年10月24日 Provider / タイプ 改定 1.1 2008年4月30日 Group 改定 1.2 2011年7月13日 Group 改定 1.3 2022年3月9日 Group / Database references / Derived calculationsカテゴリ database_2 / pdbx_nmr_software ... database_2 / pdbx_nmr_software / pdbx_nmr_spectrometer / pdbx_struct_assembly / pdbx_struct_oper_list / struct_ref_seq_dif Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_nmr_software.name / _pdbx_nmr_spectrometer.model / _struct_ref_seq_dif.details 改定 1.4 2024年5月29日 Group / カテゴリ / chem_comp_bond
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 要素
要素 キーワード
キーワード 機能・相同性情報
機能・相同性情報 Homo sapiens (ヒト)
Homo sapiens (ヒト) データ登録者
データ登録者 引用
引用 ジャーナル: To be Published
ジャーナル: To be Published 構造の表示
構造の表示 Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク ダウンロード
ダウンロード 2dmv.cif.gz
2dmv.cif.gz PDBx/mmCIF形式
PDBx/mmCIF形式 pdb2dmv.ent.gz
pdb2dmv.ent.gz PDB形式
PDB形式 2dmv.json.gz
2dmv.json.gz PDBx/mmJSON形式
PDBx/mmJSON形式 その他のダウンロード
その他のダウンロード 2dmv_validation.pdf.gz
2dmv_validation.pdf.gz wwPDB検証レポート
wwPDB検証レポート 2dmv_full_validation.pdf.gz
2dmv_full_validation.pdf.gz 2dmv_validation.xml.gz
2dmv_validation.xml.gz 2dmv_validation.cif.gz
2dmv_validation.cif.gz https://data.pdbj.org/pub/pdb/validation_reports/dm/2dmv
https://data.pdbj.org/pub/pdb/validation_reports/dm/2dmv ftp://data.pdbj.org/pub/pdb/validation_reports/dm/2dmv
ftp://data.pdbj.org/pub/pdb/validation_reports/dm/2dmv リンク
リンク 集合体
集合体
 要素
要素 Homo sapiens (ヒト) / 解説: Cell-free protein synthesis / 遺伝子: ITCH / プラスミド: P050620-31 / 発現宿主: Cell free synthesis
Homo sapiens (ヒト) / 解説: Cell-free protein synthesis / 遺伝子: ITCH / プラスミド: P050620-31 / 発現宿主: Cell free synthesis 試料調製
試料調製 解析
解析 ムービー
ムービー コントローラー
コントローラー



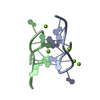



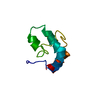
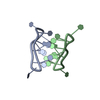
 PDBj
PDBj






 HSQC
HSQC