[English] 日本語
 Yorodumi
Yorodumi- PDB-2def: PEPTIDE DEFORMYLASE CATALYTIC CORE (RESIDUES 1-147), NMR, 20 STRU... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2def | ||||||
|---|---|---|---|---|---|---|---|
| Title | PEPTIDE DEFORMYLASE CATALYTIC CORE (RESIDUES 1-147), NMR, 20 STRUCTURES | ||||||
 Components Components | PEPTIDE DEFORMYLASE | ||||||
 Keywords Keywords | HYDROLASE / METALLOPROTEASE | ||||||
| Function / homology |  Function and homology information Function and homology informationpeptide deformylase / peptide deformylase activity / ferrous iron binding / ribosome binding / translation / hydrolase activity / zinc ion binding / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR / DISTANCE GEOMETRY, RESTRAINED SIMULATED ANNEALING | ||||||
 Authors Authors | Meinnel, T. / Dardel, F. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 1998 Journal: J.Mol.Biol. / Year: 1998Title: Solution structure of nickel-peptide deformylase. Authors: Dardel, F. / Ragusa, S. / Lazennec, C. / Blanquet, S. / Meinnel, T. #1:  Journal: FEBS Lett. / Year: 1996 Journal: FEBS Lett. / Year: 1996Title: The C-Terminal Domain of Peptide Deformylase is Disordered and Dispensable for Activity Authors: Meinnel, T. / Lazennec, C. / Dardel, F. / Schmitter, J.M. / Blanquet, S. #2:  Journal: J.Mol.Biol. / Year: 1996 Journal: J.Mol.Biol. / Year: 1996Title: A New Subclass of the Zinc Metalloproteases Superfamily Revealed by the Solution Structure of Peptide Deformylase Authors: Meinnel, T. / Blanquet, S. / Dardel, F. #3:  Journal: J.Mol.Biol. / Year: 1995 Journal: J.Mol.Biol. / Year: 1995Title: Mapping of the Active Site Zinc Ligands of Peptide Deformylase Authors: Meinnel, T. / Lazennec, C. / Blanquet, S. #4:  Journal: J.Bacteriol. / Year: 1993 Journal: J.Bacteriol. / Year: 1993Title: Evidence that Peptide Deformylase and Methionyl-tRNA(Fmet) Formyltransferase are Encoded within the Same Operon in Escherichia Coli Authors: Meinnel, T. / Blanquet, S. | ||||||
| History |
|
- Structure visualization
Structure visualization
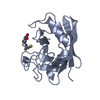
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2def.cif.gz 2def.cif.gz | 912.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2def.ent.gz pdb2def.ent.gz | 755.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2def.json.gz 2def.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/de/2def https://data.pdbj.org/pub/pdb/validation_reports/de/2def ftp://data.pdbj.org/pub/pdb/validation_reports/de/2def ftp://data.pdbj.org/pub/pdb/validation_reports/de/2def | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 16645.125 Da / Num. of mol.: 1 / Fragment: ACTIVE CATALYTIC CORE, RESIDUES 1 - 147 / Mutation: S1A Source method: isolated from a genetically manipulated source Details: ACTIVE FORM CONTAINING ONE NICKEL ION IN THE METAL BINDING SITE Source: (gene. exp.)   |
|---|---|
| #2: Chemical | ChemComp-NI / |
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Sample conditions | pH: 7.2 / Temperature: 318 K |
|---|---|
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| NMR spectrometer | Type: Bruker AMX600 & DRX600 / Manufacturer: Bruker / Model: AMX600 & DRX600 / Field strength: 600 MHz |
|---|
- Processing
Processing
| Software |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR software |
| ||||||||||||
| Refinement | Method: DISTANCE GEOMETRY, RESTRAINED SIMULATED ANNEALING / Software ordinal: 1 Details: RESTRAINED SIMULATED ANNEALING USING 1948 NOE RESTRAINTS -389 INTRARESIDUE -330 SEQUENTIAL -328 MEDIUM RANGE -901 LONG RANGE 227 DIHEDRAL ANGLE RESTRAINTS -96 PHI -13 PSI -118 CHI1 86 RESTRAINTS FOR 43 H-BONDS | ||||||||||||
| NMR ensemble | Conformer selection criteria: LOWEST DIANA TARGET FUNCTION / Conformers calculated total number: 200 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller











 PDBj
PDBj

 HSQC
HSQC