[English] 日本語
 Yorodumi
Yorodumi- PDB-2d32: Crystal Structure of Michaelis Complex of gamma-Glutamylcysteine ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2d32 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal Structure of Michaelis Complex of gamma-Glutamylcysteine Synthetase | ||||||
 Components Components | Glutamate--cysteine ligase | ||||||
 Keywords Keywords | LIGASE / glutathione homeostasis / peptide synthesis / phosphoryl transfer | ||||||
| Function / homology |  Function and homology information Function and homology informationglutamate-cysteine ligase / glutamate-cysteine ligase activity / cellular response to mercury ion / glutathione biosynthetic process / cellular response to arsenic-containing substance / hyperosmotic response / ATP binding / metal ion binding / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.4 Å MOLECULAR REPLACEMENT / Resolution: 2.4 Å | ||||||
 Authors Authors | Hibi, T. / Nakayama, M. / Nii, H. / Kurokawa, Y. / Katano, H. / Oda, J. | ||||||
 Citation Citation |  Journal: To be Published Journal: To be PublishedTitle: Structural basis of efficient coupling peptide ligation and ATP hydrolysis by gamma-gluatamylcysteine synthetase Authors: Hibi, T. / Nakayama, M. / Nii, H. / Kurokawa, Y. / Katano, H. / Oda, J. #1:  Journal: Proc.Natl.Acad.Sci.USA / Year: 2004 Journal: Proc.Natl.Acad.Sci.USA / Year: 2004Title: Crystal structure of gamma-glutamylcysteine synthetase: insights into the mechanism of catalysis by a key enzyme for glutathione homeostasis. Authors: Hibi, T. / Nii, H. / Nakatsu, T. / Kimura, A. / Kato, H. / Hiratake, J. / Oda, J. #2: Journal: Acta Crystallogr.,Sect.D / Year: 2002 Title: Escherichia coli B gamma-glutamylcysteine synthetase: modification, purification, crystallization and preliminary crystallographic analysis. Authors: Hibi, T. / Hisada, H. / Nakatsu, T. / Kato, H. / Oda, J. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2d32.cif.gz 2d32.cif.gz | 423 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2d32.ent.gz pdb2d32.ent.gz | 340.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2d32.json.gz 2d32.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/d3/2d32 https://data.pdbj.org/pub/pdb/validation_reports/d3/2d32 ftp://data.pdbj.org/pub/pdb/validation_reports/d3/2d32 ftp://data.pdbj.org/pub/pdb/validation_reports/d3/2d32 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1v4gS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 4 molecules ABCD
| #1: Protein | Mass: 58268.844 Da / Num. of mol.: 4 / Mutation: C106S, C164S, C205S, C223S Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Non-polymers , 5 types, 825 molecules 

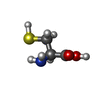






| #2: Chemical | ChemComp-MG / #3: Chemical | ChemComp-GLU / #4: Chemical | ChemComp-CYS / #5: Chemical | ChemComp-ANP / #6: Water | ChemComp-HOH / | |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 4.77 Å3/Da / Density % sol: 74 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop / pH: 7.5 Details: sodium formate, Tris-HCl, pH 7.5, VAPOR DIFFUSION, SITTING DROP, temperature 293K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SPring-8 SPring-8  / Beamline: BL38B1 / Wavelength: 0.9 Å / Beamline: BL38B1 / Wavelength: 0.9 Å |
| Detector | Type: RIGAKU / Detector: IMAGE PLATE / Date: Apr 14, 2005 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9 Å / Relative weight: 1 |
| Reflection | Resolution: 2.4→43.6 Å / Num. obs: 162223 / % possible obs: 100 % / Redundancy: 8.1 % / Biso Wilson estimate: 50.5 Å2 / Rmerge(I) obs: 0.088 / Net I/σ(I): 6.1 |
| Reflection shell | Resolution: 2.4→2.53 Å / Redundancy: 7.9 % / Rmerge(I) obs: 0.374 / Mean I/σ(I) obs: 2 / Num. unique all: 23722 / % possible all: 100 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1V4G Resolution: 2.4→40 Å / Cor.coef. Fo:Fc: 0.958 / Cor.coef. Fo:Fc free: 0.942 / SU B: 9.086 / SU ML: 0.111 / Cross valid method: THROUGHOUT / ESU R: 0.181 / ESU R Free: 0.16 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 45.908 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.276 Å / Luzzati d res low obs: 5 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.4→40 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.4→2.462 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller












 PDBj
PDBj



















