+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1zww | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of endophilin-A1 BAR domain | ||||||
 Components Components | SH3-containing GRB2-like protein 2 | ||||||
 Keywords Keywords | TRANSFERASE / coiled coil | ||||||
| Function / homology |  Function and homology information Function and homology informationmembrane bending / positive regulation of membrane tubulation / lipid tube assembly / membrane tubulation / vesicle scission / postsynaptic actin cytoskeleton organization / regulation of clathrin-dependent endocytosis / Negative regulation of MET activity / Lysosome Vesicle Biogenesis / EGFR downregulation ...membrane bending / positive regulation of membrane tubulation / lipid tube assembly / membrane tubulation / vesicle scission / postsynaptic actin cytoskeleton organization / regulation of clathrin-dependent endocytosis / Negative regulation of MET activity / Lysosome Vesicle Biogenesis / EGFR downregulation / Retrograde neurotrophin signalling / negative regulation of blood-brain barrier permeability / dendrite extension / regulation of receptor internalization / Recycling pathway of L1 / photoreceptor ribbon synapse / Golgi Associated Vesicle Biogenesis / Cargo recognition for clathrin-mediated endocytosis / Clathrin-mediated endocytosis / MHC class II antigen presentation / negative regulation of epidermal growth factor receptor signaling pathway / synaptic vesicle endocytosis / presynaptic cytosol / negative regulation of MAPK cascade / cellular response to brain-derived neurotrophic factor stimulus / hippocampal mossy fiber to CA3 synapse / Schaffer collateral - CA1 synapse / neuron projection development / synaptic vesicle membrane / presynapse / transmembrane transporter binding / early endosome / postsynapse / negative regulation of gene expression / lipid binding / synapse / protein kinase binding / perinuclear region of cytoplasm / glutamatergic synapse / identical protein binding / cytoplasm Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  SAD / Resolution: 2.3 Å SAD / Resolution: 2.3 Å | ||||||
 Authors Authors | Weissenhorn, W. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2005 Journal: J.Mol.Biol. / Year: 2005Title: Crystal Structure of the Endophilin-A1 BAR Domain. Authors: Weissenhorn, W. | ||||||
| History |
|
- Structure visualization
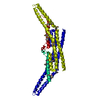
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1zww.cif.gz 1zww.cif.gz | 91.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1zww.ent.gz pdb1zww.ent.gz | 68.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1zww.json.gz 1zww.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/zw/1zww https://data.pdbj.org/pub/pdb/validation_reports/zw/1zww ftp://data.pdbj.org/pub/pdb/validation_reports/zw/1zww ftp://data.pdbj.org/pub/pdb/validation_reports/zw/1zww | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 29339.547 Da / Num. of mol.: 2 / Fragment: endophilin BAR domain Source method: isolated from a genetically manipulated source Source: (gene. exp.)   References: UniProt: Q62420, Transferases; Acyltransferases; Transferring groups other than aminoacyl groups #2: Chemical | ChemComp-CD / #3: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.6 Å3/Da / Density % sol: 52.4 % |
|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, hanging drop / pH: 9 Details: sodium acetate, cadmium sulfate, VAPOR DIFFUSION, HANGING DROP, temperature 298K, pH 9.00 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: BM14 / Wavelength: 0.97879 / Beamline: BM14 / Wavelength: 0.97879 |
| Detector | Date: Mar 14, 2005 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.97879 Å / Relative weight: 1 |
| Reflection | Resolution: 2.3→48.7 Å / Num. obs: 27463 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  SAD / Resolution: 2.3→20 Å / Cor.coef. Fo:Fc: 0.928 / Cor.coef. Fo:Fc free: 0.913 / SU B: 6.459 / SU ML: 0.161 / Cross valid method: THROUGHOUT / ESU R: 0.249 / ESU R Free: 0.231 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS SAD / Resolution: 2.3→20 Å / Cor.coef. Fo:Fc: 0.928 / Cor.coef. Fo:Fc free: 0.913 / SU B: 6.459 / SU ML: 0.161 / Cross valid method: THROUGHOUT / ESU R: 0.249 / ESU R Free: 0.231 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 44.03 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.3→20 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.3→2.36 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller










 PDBj
PDBj








