+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1xc0 | ||||||
|---|---|---|---|---|---|---|---|
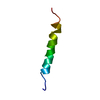
| Title | Twenty Lowest Energy Structures of Pa4 by Solution NMR | ||||||
 Components Components | Pardaxin P-4 | ||||||
 Keywords Keywords | SIGNALING PROTEIN / Bend-helix-bend-helix motif | ||||||
| Function / homology | Pardaxin / Pardaxin / other organism cell membrane / monoatomic ion transport / toxin activity / extracellular region / Pardaxin P-4 Function and homology information Function and homology information | ||||||
| Method | SOLUTION NMR / extended structure; random simulated annealing; molecular dynamics | ||||||
 Authors Authors | Porcelli, F. / Buck, B. / Lee, D.-K. / Hallock, K.J. / Ramamoorthy, A. / Veglia, G. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2004 Journal: J.Biol.Chem. / Year: 2004Title: Structure and orientation of pardaxin determined by NMR experiments in model membranes Authors: Porcelli, F. / Buck, B. / Lee, D.-K. / Hallock, K.J. / Ramamoorthy, A. / Veglia, G. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1xc0.cif.gz 1xc0.cif.gz | 207.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1xc0.ent.gz pdb1xc0.ent.gz | 174.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1xc0.json.gz 1xc0.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/xc/1xc0 https://data.pdbj.org/pub/pdb/validation_reports/xc/1xc0 ftp://data.pdbj.org/pub/pdb/validation_reports/xc/1xc0 ftp://data.pdbj.org/pub/pdb/validation_reports/xc/1xc0 | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein/peptide | Mass: 3325.851 Da / Num. of mol.: 1 / Source method: obtained synthetically Details: Pardaxins naturally occur in sole fish of genus Pardachirus. Pa4 was synthesized using standard 9-fluorenylmethoxycarbonyl-based solid-pahse methods with an ABI 431A peptide synthesizer. ...Details: Pardaxins naturally occur in sole fish of genus Pardachirus. Pa4 was synthesized using standard 9-fluorenylmethoxycarbonyl-based solid-pahse methods with an ABI 431A peptide synthesizer. Crude peptide was purified by reversed-phase HPLC References: UniProt: P81861 |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Details | Contents: ~1.5 mg of lyophilized Pa4 in 20 mM PBS (pH 6.5),300 mM d31-DPC and 10% D2O. Sample pH was adjusted to ~4.5 with NaOH. Solvent system: 90% water and 10% D2O |
|---|---|
| Sample conditions | pH: 4.5 / Temperature: 303 K |
-NMR measurement
| NMR spectrometer | Type: Varian INOVA / Manufacturer: Varian / Model: INOVA / Field strength: 800 MHz |
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: extended structure; random simulated annealing; molecular dynamics Software ordinal: 1 / Details: 335 NOEs; 13 H-bonds | ||||||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the lowest energy Conformers calculated total number: 350 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller



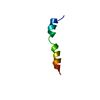








 PDBj
PDBj NMRPipe
NMRPipe