[English] 日本語
 Yorodumi
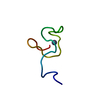
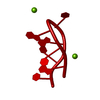
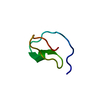
Yorodumi- PDB-1med: METHIONYL-TRNA SYNTHETASE ZINC BINDING DOMAIN. 3D STRUCTURE AND H... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1med | ||||||
|---|---|---|---|---|---|---|---|
| Title | METHIONYL-TRNA SYNTHETASE ZINC BINDING DOMAIN. 3D STRUCTURE AND HOMOLOGY WITH RUBREDOXIN AND GAG RETROVIRAL PROTEINS | ||||||
 Components Components | METHIONYL-tRNA SYNTHETASE | ||||||
 Keywords Keywords | AMINOACYL-TRNA SYNTHASE | ||||||
| Function / homology |  Function and homology information Function and homology informationmethionine-tRNA ligase / methionine-tRNA ligase activity / methionyl-tRNA aminoacylation / cellular response to antibiotic / tRNA binding / protein homodimerization activity / zinc ion binding / ATP binding / membrane / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR | ||||||
 Authors Authors | Fourmy, D. / Dardel, F. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 1993 Journal: J.Mol.Biol. / Year: 1993Title: Methionyl-tRNA synthetase zinc binding domain. Three-dimensional structure and homology with rubredoxin and gag retroviral proteins. Authors: Fourmy, D. / Dardel, F. / Blanquet, S. #1:  Journal: J.Mol.Biol. / Year: 1993 Journal: J.Mol.Biol. / Year: 1993Title: Mapping of the Zinc Binding Domain of Escherichia Coli Methionyl-tRNA Synthetase Authors: Fourmy, D. / Meinnel, T. / Mechulam, Y. / Blanquet, S. #2:  Journal: J.Mol.Biol. / Year: 1990 Journal: J.Mol.Biol. / Year: 1990Title: Crystallographic Study at 2.5 Angstroms Resolution of the Interaction of Methionyl-tRNA Synthetase from Escherichia Coli with ATP Authors: Brunie, S. / Zelwer, C. / Risler, J.-L. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1med.cif.gz 1med.cif.gz | 93 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1med.ent.gz pdb1med.ent.gz | 57.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1med.json.gz 1med.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/me/1med https://data.pdbj.org/pub/pdb/validation_reports/me/1med ftp://data.pdbj.org/pub/pdb/validation_reports/me/1med ftp://data.pdbj.org/pub/pdb/validation_reports/me/1med | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| Atom site foot note | 1: CYS 10 - PRO 11 MODEL 1 OMEGA =221.65 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 2: SER 15 - PRO 16 MODEL 1 OMEGA =230.63 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 3: CYS 10 - PRO 11 MODEL 2 OMEGA =226.31 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 4: SER 15 - PRO 16 MODEL 2 OMEGA =233.15 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 5: CYS 10 - PRO 11 MODEL 4 OMEGA =212.66 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 6: SER 15 - PRO 16 MODEL 4 OMEGA =223.41 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 7: CYS 10 - PRO 11 MODEL 5 OMEGA =227.82 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 8: CYS 10 - PRO 11 MODEL 6 OMEGA =216.14 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 9: SER 15 - PRO 16 MODEL 6 OMEGA =213.88 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 10: CYS 10 - PRO 11 MODEL 8 OMEGA =211.33 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 11: SER 15 - PRO 16 MODEL 8 OMEGA =214.66 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 12: CYS 10 - PRO 11 MODEL 9 OMEGA =215.05 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 13: SER 15 - PRO 16 MODEL 9 OMEGA =234.13 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 14: CYS 10 - PRO 11 MODEL 10 OMEGA =221.52 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 15: SER 15 - PRO 16 MODEL 10 OMEGA =220.78 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 16: CYS 10 - PRO 11 MODEL 11 OMEGA =212.67 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 17: SER 15 - PRO 16 MODEL 11 OMEGA =236.00 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION | |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein/peptide | Mass: 2969.311 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  |
|---|---|
| #2: Chemical | ChemComp-ZN / |
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR |
|---|
- Processing
Processing
| Software |
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| NMR ensemble | Conformers submitted total number: 11 |
 Movie
Movie Controller
Controller




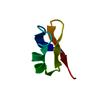


 PDBj
PDBj


 X-PLOR
X-PLOR